P olicies surrounding precise measurements in chemical compounds are pivotal for various industries, including pharmaceuticals and materials science. A fundamental aspect of such precision is understanding the exact caf2 molar mass. The term molar mass, representing the mass of a given substance (chemical element or chemical compound) divided by the amount of substance (in moles), is critical in these fields. In this article, we dive deep into the precise calculation of caf2 molar mass and explore its broader implications.
Understanding the Basics: What is Caf2?
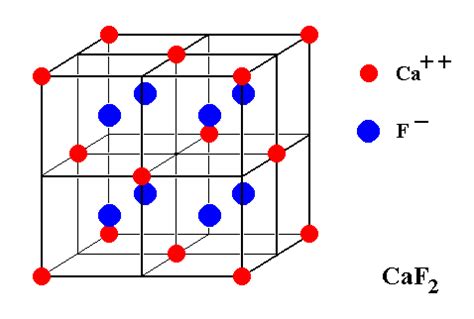
Caf2, or calcium fluoride, is an ionic compound consisting of calcium (Ca) and fluorine (F) atoms. It is widely recognized for its applications in optics, specifically in the production of lenses and optical components due to its excellent transparency and low dispersion properties. The accurate determination of the caf2 molar mass is crucial for formulating precise quantities needed in manufacturing processes.Key Insights
Key Insights
- Primary insight with practical relevance: Accurate determination of the caf2 molar mass is essential for manufacturing high-precision optical components.
- Technical consideration with clear application: Knowledge of the caf2 molar mass aids in accurately predicting material behavior under various conditions.
- Actionable recommendation: Employ high-precision instruments and validated analytical methods to determine the caf2 molar mass for optimal industrial applications.
Calculating the Caf2 Molar Mass
To accurately calculate the molar mass of caf2, we need to understand the atomic weights of calcium and fluorine. According to the periodic table, the atomic weight of calcium is approximately 40.08 g/mol, and that of fluorine is about 18.998 g/mol. Given that caf2 comprises one calcium atom and two fluorine atoms, the molar mass calculation follows:Molar mass of caf2 = Atomic weight of Ca + (2 x Atomic weight of F)
Molar mass of caf2 = 40.08 g/mol + (2 x 18.998 g/mol) = 40.08 g/mol + 37.996 g/mol = 78.076 g/mol
Implications of Accurate Molar Mass Calculation
Accurate measurement of the caf2 molar mass directly impacts several facets of the material’s application, particularly in optical technologies. A minor miscalculation can lead to inconsistencies in material properties, ultimately affecting the performance of optical devices. Moreover, in pharmaceutical formulations, precise molar mass calculations ensure the correct dosage and efficacy of calcium-containing compounds. Understanding the precise molar mass also allows scientists to tailor synthesis processes to produce caf2 in desired purity levels, thereby enhancing the quality of end products.FAQ Section
What is the significance of knowing the exact molar mass of caf2?
Knowing the exact molar mass of caf2 is crucial for ensuring the accuracy and reliability of optical devices, particularly in high-precision applications like lens manufacturing. Accurate molar mass determination also supports precise pharmaceutical formulations involving calcium compounds.
How does the molar mass of caf2 affect its use in optics?
The molar mass of caf2 influences its density, crystal structure, and optical properties such as refractive index and dispersion. Precise molar mass calculations help predict and optimize these properties, ensuring the optimal performance of optical components.
Understanding the exact caf2 molar mass is a cornerstone in both material science and industrial applications. Through precise measurement and calculation, industries can achieve the reliability and quality essential for producing advanced optical components and pharmaceuticals. Employing high-precision analytical techniques ensures that the molar mass is determined accurately, facilitating better material performance and overall efficiency in the manufacturing processes.