Nitrates in water bodies have long intrigued scientists and environmental experts alike due to their profound implications on ecosystem health, agricultural productivity, and human health. Understanding the charge of nitrate, its sources, and its environmental impact forms a crucial component in tackling related issues.
This article aims to decode the mysteries surrounding nitrates, presenting practical insights and evidence-based information. We will delve into the chemical nature of nitrates, their environmental behavior, and strategies to mitigate their adverse effects.
Key Insights
- Nitrates possess a negative charge, playing a critical role in various environmental processes.
- Understanding nitrate dynamics is essential for managing water quality and agricultural runoff.
- Implementing best management practices can significantly reduce nitrate pollution.
Chemical Nature of Nitrates
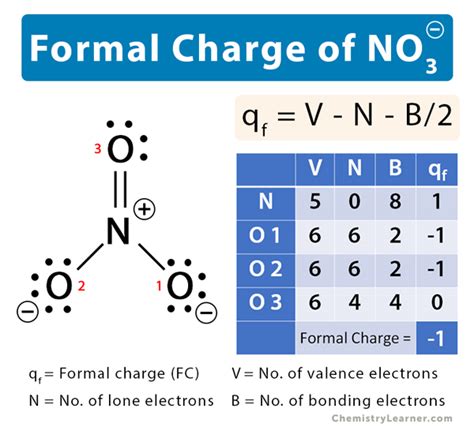
Nitrates (NO3-) are charged particles derived from nitrogen oxides. Chemically, nitrates feature a central nitrogen atom bonded to three oxygen atoms in a linear structure. The negative charge of nitrates arises due to the extra electron in the nitrogen-oxygen structure. This charge influences nitrate’s solubility in water and its mobility in soil, making it a key player in nutrient cycles within ecosystems.
Environmental Behavior of Nitrates
The behavior of nitrates in the environment can have significant ecological ramifications. Once in water bodies, nitrates can promote excessive growth of algae through a process known as eutrophication. This often leads to hypoxic conditions that severely affect aquatic life. Moreover, nitrate leaching into groundwater poses a threat to drinking water quality. Nitrate contamination in drinking water can cause methemoglobinemia, or “blue baby syndrome,” in infants. Thus, comprehending nitrate dynamics is crucial for both environmental stewardship and public health.
Nitrates primarily originate from agricultural activities, such as the application of fertilizers, and urban runoff carrying various anthropogenic pollutants. In agricultural settings, over-application of nitrogen-based fertilizers increases soil nitrate levels, facilitating nitrate leaching into groundwater. Consequently, understanding the best practices in nitrogen fertilizer management is vital for minimizing nitrate pollution.
How do we measure nitrate levels in water?
Nitrate concentrations in water can be measured using methods such as ion chromatography or spectrophotometric analysis. Regular monitoring of nitrate levels is crucial for water quality management, ensuring compliance with environmental and health standards.
What are the most effective ways to reduce nitrate runoff?
Effective strategies include optimizing fertilizer application through precision agriculture, employing cover crops to minimize soil erosion, and constructing buffer strips of vegetation along water bodies to filter runoff before it reaches water sources.
The charge of nitrate and its pervasive impact on environmental health and human well-being underscores the need for informed, strategic approaches. Through understanding the chemistry and behavior of nitrates, we can develop effective strategies to manage their presence in our ecosystems, ensuring a balanced approach between agricultural productivity and environmental sustainability.