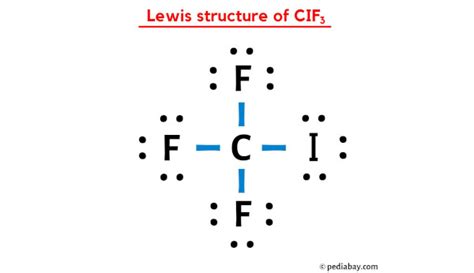
Understanding the CIF3 Lewis structure is pivotal in mastering advanced chemistry concepts. The study of molecular geometry, electron behavior, and valence electrons is significantly enhanced through this exploration. The CIF3 molecule is unique due to its three fluorine atoms and one iodine atom, creating a complex electron distribution pattern. This article delves into the intricacies of CIF3’s Lewis structure, providing practical insights and evidence-based statements.
Key Insights
- CIF3 features a central iodine atom bonded to three fluorine atoms, with an unshared pair on iodine.
- Its molecular geometry is T-shaped due to the presence of a lone pair on iodine.
- Understanding CIF3's structure aids in grasping hypervalent molecules' behavior.
Electron Configuration and Bonding in CIF3
CIF3 showcases the concept of hypervalency, a phenomenon where an atom exhibits more than eight electrons in its valence shell. The iodine atom in CIF3, being a member of Group 17, typically forms a maximum of two bonds in simpler molecules. However, in CIF3, iodine forms three bonds with fluorine atoms, supplemented by a lone pair, leading to a total of ten electrons around iodine. This hypervalent structure results from the availability of d-orbitals in iodine’s electron configuration, allowing it to expand its octet.Molecular Geometry and Spatial Arrangement
The spatial arrangement of CIF3 can be described through its molecular geometry, which is T-shaped. This geometry arises due to the repulsion between the three bonding pairs and one lone pair of electrons surrounding the central iodine atom. According to the VSEPR (Valence Shell Electron Pair Repulsion) theory, the lone pair occupies more spatial volume than the bonding pairs, thus influencing the shape of the molecule. The bonding pairs between iodine and each fluorine atom align to minimize repulsion, resulting in the characteristic T-shape of CIF3.To illustrate, consider the positioning of the fluorine atoms. Two are positioned in axial locations to maximize distance from each other and the lone pair, while the third occupies one of the equatorial positions to optimize its interaction with the other atoms. This arrangement not only satisfies the steric demands but also adheres to the octet rule for fluorine atoms, completing their electron requirements with minimal repulsion.
How does the presence of a lone pair affect CIF3's properties?
The lone pair on the iodine atom contributes to CIF3's unique reactivity and T-shaped geometry. It significantly influences the molecule's polarity, affecting its chemical behavior and interactions in various environments.
Why is CIF3 considered hypervalent?
CIF3 is hypervalent because it features more than eight electrons around the central iodine atom. This hypervalency is made possible by the use of iodine's d-orbitals in bonding, allowing for an expanded octet beyond the conventional rule.
Understanding CIF3’s Lewis structure is essential for appreciating the complexities of hypervalent molecules. Through this exploration, we gain insights into molecular geometry, electron distribution, and the broader implications for chemical reactivity. This knowledge is not only academically enriching but also practically significant for fields like medicinal chemistry and material science where hypervalent compounds play a crucial role.