Mastering CN Lewis Structure: Quick and Accurate Guide for Chemistry Enthusiasts
In the realm of chemistry, understanding Lewis structures is vital for comprehending the electron configuration and bonding of molecules. The cyanide ion (CN⁻) is an especially intriguing molecule due to its unusual structure and electronic properties. Here, we present a succinct, evidence-based guide to accurately drawing the CN Lewis structure, providing practical insights for chemistry enthusiasts.
Key Insights
- Cyanide (CN⁻) has a total of 10 valence electrons to distribute.
- Understanding the central atom (Carbon or Nitrogen) is crucial for proper bonding.
- Properly distributing electrons ensures that both carbon and nitrogen follow the octet rule.
Understanding Valence Electrons
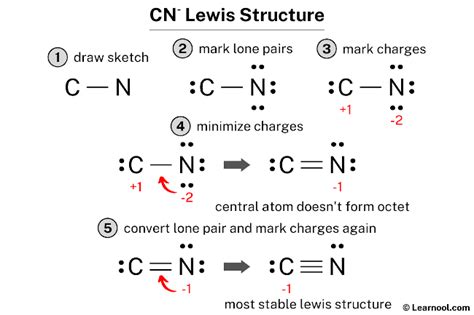
The first step in drawing a CN Lewis structure is to determine the total valence electrons. Carbon has four valence electrons, while nitrogen contributes five. Additionally, the cyanide ion has one extra electron due to its -1 charge, resulting in a total of ten valence electrons. Recognizing the electron count forms the foundation for constructing an accurate Lewis structure.
Determining Central Atom and Bond Formation
The CN molecule presents a decision on the central atom. Typically, nitrogen, being more electronegative, tends to be the central atom. In CN⁻, nitrogen can form three bonds with a lone pair of electrons, while carbon can complete its octet by forming a triple bond with nitrogen. This arrangement satisfies the octet rule for both atoms, ensuring a stable structure.
The triple bond between carbon and nitrogen is not merely a figure of speech; it reflects a triple covalent bond where three pairs of electrons are shared between the two atoms. This bond type provides stability and distinct reactivity for the CN⁻ ion.
What is the difference between a single, double, and triple bond in terms of electron sharing?
In a single bond, two pairs of electrons are shared between atoms; in a double bond, four pairs; and in a triple bond, six pairs are shared. These differences impact bond length, strength, and the molecule's overall properties.
How does the charge of the cyanide ion affect its Lewis structure?
The -1 charge in CN⁻ adds an extra electron to the total valence electron count. This extra electron is distributed in the Lewis structure as a lone pair on the nitrogen atom, ensuring all atoms achieve the octet rule.
This guide aims to streamline the process of mastering the CN Lewis structure for chemistry enthusiasts, ensuring accurate and efficient comprehension. As with any molecular structure, practice and understanding of fundamental principles are key. By adhering to these guidelines, students and professionals alike can deepen their grasp of Lewis structures and their applications in various chemical processes.