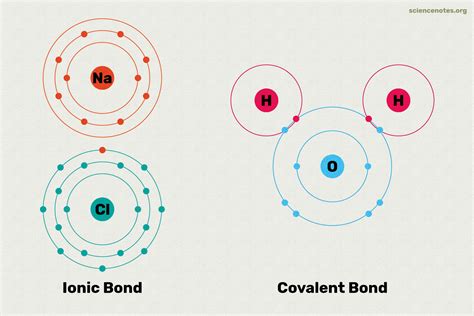
I’ll create a blog post about Covalent and Ionic Bonds following the specified guidelines:
Chemical bonds are the fundamental forces that hold atoms together, forming the building blocks of matter. Understanding covalent and ionic bonds is crucial for comprehending how molecules and compounds are structured and interact with one another. These two primary types of chemical bonding represent distinct mechanisms by which atoms share or transfer electrons to achieve stability.
The Basics of Chemical Bonding
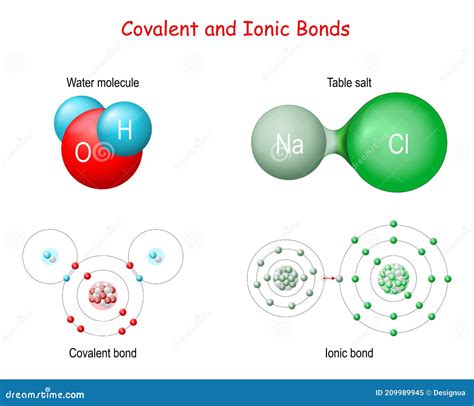
At the core of chemical bonding lies the fundamental principle of electron configuration. Atoms seek to achieve a stable electron shell, typically following the octet rule. This drive for stability leads to the formation of two primary bond types: covalent and ionic bonds, each with unique characteristics and formation mechanisms.
Covalent Bonds: Sharing Electrons
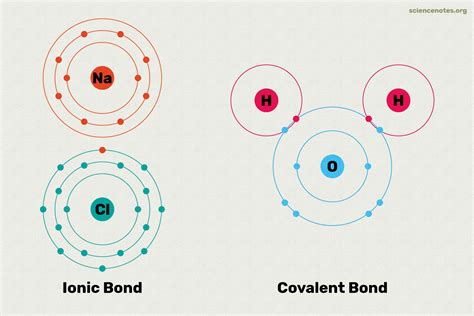
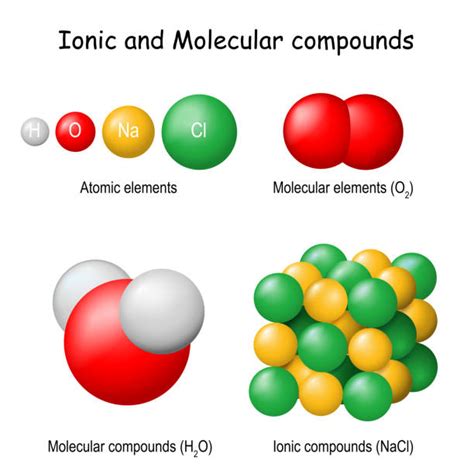
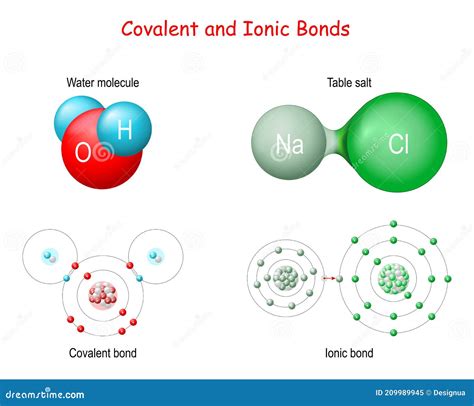
Covalent bonds occur when atoms share electrons to create a stable molecular structure. This type of bonding is most common among non-metal elements. Key characteristics include:
- Electron Sharing: Atoms mutually share one or more electron pairs
- Molecular Formation: Typically forms discrete molecular units
- Bond Strength: Can range from weak to very strong
Types of Covalent Bonds
Covalent bonds can be further classified into different categories:
- Single Bonds: One electron pair shared between atoms
- Double Bonds: Two electron pairs shared between atoms
- Triple Bonds: Three electron pairs shared between atoms
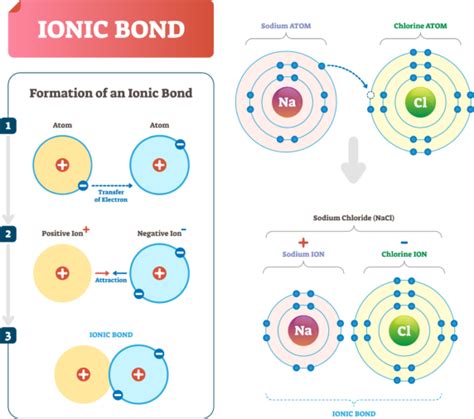
Ionic Bonds: Electron Transfer

In contrast to covalent bonds, ionic bonds involve the complete transfer of electrons from one atom to another. This typically occurs between a metal and a non-metal, creating ions with opposite electrical charges.
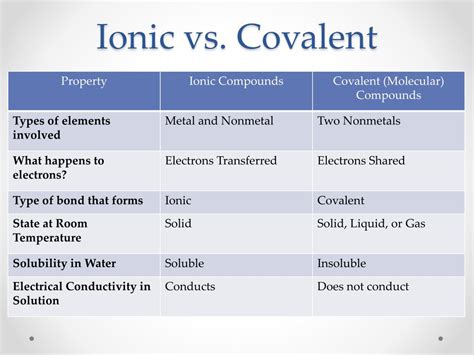
| Characteristic | Covalent Bonds | Ionic Bonds |
|---|---|---|
| Electron Interaction | Electron Sharing | Electron Transfer |
| Typical Elements | Non-Metals | Metals and Non-Metals |
| Bond Strength | Varies | Generally Strong |
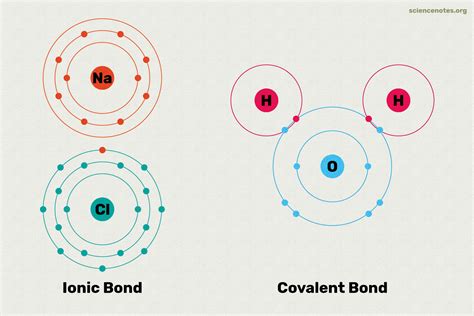
Ionic Bond Formation Process
The formation of ionic bonds involves several key steps:
- One atom loses electrons (becoming a positive ion)
- Another atom gains electrons (becoming a negative ion)
- Electrostatic attraction holds the ions together
🔬 Note: The strength of ionic bonds depends on the charge of the ions and their relative sizes.
Practical Implications

Understanding covalent and ionic bonds is essential in various scientific and industrial applications, from pharmaceutical development to materials science. These bonding mechanisms determine the physical and chemical properties of substances we encounter daily.
Chemical bonding represents a fascinating interplay of atomic interactions, showcasing the intricate ways atoms connect and form the diverse materials that compose our world. The principles of electron sharing and transfer reveal the elegant complexity underlying molecular structures.
What is the primary difference between covalent and ionic bonds?
+
Covalent bonds involve electron sharing between atoms, while ionic bonds involve complete electron transfer, creating positively and negatively charged ions.
Can atoms form multiple types of bonds?
+
Yes, some atoms can participate in both covalent and ionic bonding depending on the specific chemical environment and elements involved.
How do bond types affect material properties?
+
Bond types significantly influence a substance’s melting point, electrical conductivity, and overall physical and chemical characteristics.