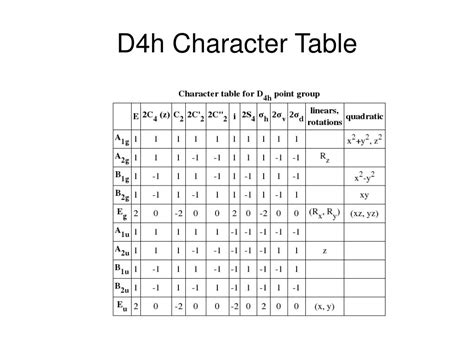
Understanding the D4H character table is crucial for anyone involved in computational chemistry or spectroscopy. This table reveals how the symmetry of a molecule impacts its electronic structure, vibrational modes, and spectroscopic properties. This knowledge is essential for both academic research and practical applications in material science. By mastering the D4H character table, chemists can predict the behavior of complex molecules under various conditions, leading to advancements in fields such as pharmaceuticals and nanotechnology.
Key Insights
- The D4H character table helps determine which vibrational modes are infrared active and Raman active.
- Understanding symmetry elements like planes and axes in D4H symmetry can simplify complex calculations.
- Using the D4H character table can lead to more accurate predictions in spectroscopy and molecular behavior.
The Importance of Symmetry in D4H
The D4H point group is characterized by its four rotational axes and various reflection planes. Recognizing these symmetry elements allows chemists to classify molecular vibrations. This classification is vital for interpreting experimental data from infrared and Raman spectroscopy. For instance, vibrations corresponding to irreducible representations that include A1 or A2 will be active in both infrared and Raman spectra if they have a change in the dipole moment or polarizability.Vibrational Modes and Spectroscopic Activity
To determine which vibrational modes are spectroscopically active, one must refer to the D4H character table. This table maps out how each vibration behaves under the different symmetry operations. Modes belonging to the irreducible representations A1, A2, B1, B2, E, and F2 are all possible in this symmetry group. For example, modes labeled E will be doubly degenerate and will either involve in-phase or out-of-phase movements along the two-fold axes. Knowing these patterns is critical for accurately predicting how a molecule will interact with light and, thus, how it will be detected in spectroscopic studies.When interpreting the results, it is important to consider the specific physical properties that each vibrational mode influences. Modes designated as F2 often relate to changes in polarizability rather than dipole moment, which means they appear in Raman spectra but not necessarily in infrared. This distinction helps chemists design experiments that can reveal the full spectrum of molecular interactions.
What is the difference between infrared and Raman active modes in D4H symmetry?
Infrared active modes are characterized by a change in the dipole moment during vibration, making them detectable in IR spectroscopy. Raman active modes involve changes in the polarizability of the molecule, which can be detected via Raman spectroscopy. In D4H symmetry, modes labeled as A1, A2, B1, B2, E, and F2 can all potentially be either infrared or Raman active depending on the specific nature of the vibration.
How does the D4H character table aid in computational chemistry?
The D4H character table aids computational chemists by providing a framework to predict the symmetry of molecular vibrations. This prediction can simplify computational models by focusing on relevant degrees of freedom and by understanding how vibrational modes will interact with electromagnetic radiation, thus enhancing the accuracy of spectroscopic predictions.
The mastery of the D4H character table enables chemists to decode the complexities of molecular behavior, leading to innovative advancements in scientific research and industrial applications. The intersection of symmetry and spectroscopy opens up a wealth of possibilities for exploring and manipulating molecular structures, driving progress across many scientific domains.