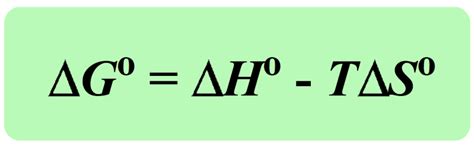Pinning down the nuances of chemical reactions is paramount for both academic and industrial chemistry fields. Understanding the master delta G formula is crucial for determining the spontaneity of a reaction. The Gibbs free energy change (ΔG) provides insight into whether a chemical process will proceed without external intervention. This article delves into the core principles behind the master delta G formula, offering practical insights and real-world examples.
Key Insights
- ΔG indicates the spontaneity of a reaction: a negative ΔG implies a spontaneous process.
- ΔG calculation includes both enthalpy and entropy changes; ΔG = ΔH - TΔS.
- Monitoring ΔG helps in optimizing industrial chemical processes for maximum efficiency.
Fundamentals of Gibbs Free Energy
The Gibbs free energy change (ΔG) is a thermodynamic quantity that measures the maximum reversible work that may be performed by a thermodynamic system at constant temperature and pressure. It’s given by the equation: ΔG = ΔH - TΔS, where ΔH represents the change in enthalpy, T is the absolute temperature in Kelvin, and ΔS represents the change in entropy. This formula elegantly captures both the energy (enthalpy) and disorder (entropy) factors critical for understanding the spontaneity of a reaction.
Interplay of Enthalpy and Entropy
To grasp the master delta G formula, one must first appreciate the role of enthalpy and entropy. Enthalpy change (ΔH) relates to the heat content of the system, while entropy change (ΔS) pertains to the degree of disorder or randomness. In exothermic reactions, ΔH is negative, contributing to a negative ΔG. Conversely, in endothermic reactions, ΔH is positive, often requiring external energy input. However, entropy changes are equally vital: a positive ΔS can offset a positive ΔH, resulting in a negative ΔG and a spontaneous process.
Consider a real-world example: the industrial synthesis of ammonia via the Haber process. Here, ΔG for the reaction is negative at high pressure and low temperature, ensuring the process’s spontaneity and efficiency.
Can ΔG ever be positive in a spontaneous reaction?
No, a positive ΔG signifies that a reaction is nonspontaneous under given conditions. However, by changing conditions like temperature or pressure, ΔG can shift, potentially making the reaction feasible.
Why is ΔG important in industrial chemistry?
ΔG is crucial for optimizing industrial processes. By calculating ΔG, chemists can predict and manipulate reaction conditions to maximize yield and efficiency, thus saving resources and reducing costs.
Mastering the delta G formula is not just an academic exercise but a practical tool with significant implications across various fields, from pharmaceuticals to materials science. By integrating both enthalpy and entropy, this formula enables scientists to precisely gauge reaction spontaneity, driving innovation and efficiency.