Understanding hydroxide chemistry can feel like deciphering a complex scientific code, but fear not! With this guide, you will uncover practical and actionable advice to master the essentials. From identifying key concepts to tackling common challenges, we’ll equip you with the knowledge you need to navigate hydroxide chemistry successfully.
Introduction: Unveiling the Mystery of Hydroxide Chemistry
Hydroxide chemistry, while complex, is a fascinating aspect of chemical science. This guide will break down the essentials into digestible, actionable steps. Whether you're a student, a professional chemist, or just curious, this guide offers real-world examples and practical solutions to common problems you may encounter.
Addressing Your Key Concerns: What Problems Will You Solve?
The journey into hydroxide chemistry often starts with questions about reactivity, stability, and the fundamental properties of hydroxides. We'll tackle these concerns by:
- Deciphering the behavior of hydroxide ions in various chemical environments.
- Understanding common pitfalls and how to avoid them.
- Offering practical tips to optimize your approach in hydroxide chemistry experiments.
Quick Reference: Your Immediate Action Plan
Quick Reference
- Immediate action item: Start by understanding the structure of hydroxide (OH-) to grasp its reactivity and role in different chemical reactions.
- Essential tip: Familiarize yourself with basic hydroxides like NaOH and KOH, and observe their behavior in aqueous solutions.
- Common mistake to avoid: Don't confuse hydroxide ions with hydronium ions (H3O+); their roles in reactions are fundamentally different.
Detailed How-To Sections: Dive Into Key Concepts
How to Understand Hydroxide Ion Structure
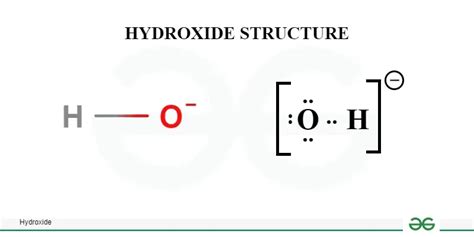
To grasp the behavior of hydroxide ions, understanding their basic structure is essential. The hydroxide ion (OH-) consists of one oxygen atom covalently bonded to one hydrogen atom with a lone pair of electrons. This structure gives hydroxide ions a negative charge. When you visualize this structure, it becomes clear why they are such powerful bases and why they readily donate a proton to acids in neutralization reactions.
Practical example: In an aqueous solution, when sodium hydroxide (NaOH) is dissolved, it dissociates into Na+ and OH- ions. The OH- ions react with any available protons from the water to form water molecules. This behavior exemplifies how hydroxide ions act in a base-neutralizing environment.
How to Identify Hydroxide Reactivity
Hydroxide ions’ reactivity is largely determined by their ability to accept protons. This property makes them strong bases and essential in various chemical processes, including titrations, neutralizations, and more.
Step-by-step guidance:
- Identify the presence of hydroxide ions in a solution by testing its pH. A basic pH indicates the presence of hydroxide ions.
- Observe neutralization reactions where acids (H+ donors) interact with hydroxide ions to form water. This reaction is central to understanding hydroxide's role in chemical equilibria.
- Experiment with different bases containing hydroxide ions to see how their strength varies based on the cation they pair with. For instance, compare NaOH and Ca(OH)2 to understand the differences in their basicity.
How to Optimize Hydroxide Experiments
To ensure success in hydroxide chemistry experiments, optimization is key. Follow these guidelines to enhance your results:
- Preparation: Always ensure that your chemicals are pure and your equipment is clean.
- Stoichiometry: Use precise measurements to avoid excess reagents that could complicate the reaction.
- Temperature control: Many hydroxide reactions are temperature-sensitive; maintain consistent temperature conditions.
For instance, when performing a titration involving a hydroxide and an acid, ensure the burette is correctly calibrated and the temperature of the solutions is stable. This will lead to more accurate results.
Practical FAQ: Your Go-To Solutions
What are common mistakes beginners make in hydroxide chemistry?
Beginners often confuse hydroxide ions with other species like hydronium ions (H3O+). It’s crucial to remember that hydroxide ions (OH-) are basic and will accept protons, whereas hydronium ions are acidic and donate protons. Another common error is not controlling the pH of the solution properly, which can skew results in experiments. To avoid this, always calibrate your pH meter and ensure that your solutions are at the expected pH before starting your experiment.
How can I improve my titration results with hydroxide?
To improve your titration results with hydroxide, follow these steps:
- Use a high-quality indicator that changes color at the expected pH for neutralization.
- Ensure that both your titrant and analyte are at room temperature to avoid temperature-induced reaction rate discrepancies.
- Take multiple titration trials to average out any inaccuracies from the first attempt.
- Double-check all measurements and calculations for accuracy.
Advanced Tips and Best Practices
For those ready to elevate their hydroxide chemistry expertise, here are advanced tips and best practices:
- Advanced preparation: Use ultrapure water and reagents to avoid impurities that could interfere with reactions.
- Precision instruments: Employ advanced analytical instruments like pH meters, burettes, and titration systems for precise control and measurement.
- Research and innovation: Stay updated with the latest research on hydroxide applications and innovative uses in industry and academia.
By following these steps, you'll not only understand but also apply hydroxide chemistry with greater confidence and skill.
This guide is designed to evolve with your understanding and skills, providing you with the foundational knowledge and advanced techniques you need to excel in hydroxide chemistry. Dive in and start mastering this fascinating field today!