The science of temperature regulation and thermal dynamics has been a cornerstone of both natural phenomena and engineered systems. Freezing point temperatures, specifically at Fahrenheit scale, often stand as a pivotal aspect of our daily lives and technological applications. Understanding the science behind what happens at 32 degrees Fahrenheit not only unveils critical natural processes but also equips us with the knowledge to innovate and improve upon various engineering solutions.
Key Insights
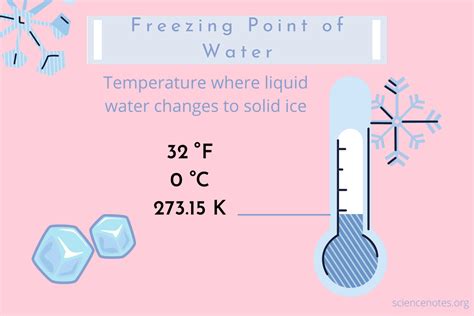
- Primary insight with practical relevance: The freezing point of water at 32°F (0°C) is crucial for both environmental and technological applications, affecting everything from climate science to refrigeration systems.
- Technical consideration with clear application: Precise control of temperatures around the freezing point is vital in pharmaceuticals, preserving biologics, and maintaining the integrity of frozen food.
- Actionable recommendation: Employ advanced cryogenic technologies to manage and store materials at temperatures close to the freezing point for extended preservation.
Understanding the exact point at which water transitions from liquid to solid is paramount in multiple scientific fields. At precisely 32°F, the molecular structure of water undergoes a phase transition where intermolecular forces dominate, and the molecules arrange into a crystalline lattice typical of ice. This seemingly simple change in state has monumental implications for climate and weather patterns. For instance, lakes and rivers freezing over can dramatically alter local ecosystems, affecting fish populations and migratory bird patterns. Additionally, the phenomenon of freezing is leveraged in various industries, such as in the food preservation industry where controlled freezing prevents bacterial growth and preserves nutritional value.
Moreover, the freezing point of water at Fahrenheit scale holds significant relevance in cryogenics, the study of matter at very low temperatures. This area of science has revolutionized the way we store biological samples, including cells, tissues, and entire embryos. Cryogenic technologies allow these samples to be preserved in a state close to freezing, maintaining their viability and function. This is particularly critical in medical research, where preserving human tissues can lead to breakthroughs in regenerative medicine.
At 32°F, water transitions from a fluid to a solid state, and this change has vast implications beyond mere observation. In engineering, understanding these principles aids in the design of systems that manage temperature, such as refrigerators, freezers, and air conditioning units. For example, refrigeration systems must accurately control the temperature around the freezing point to ensure the proper functioning of frozen goods, thus maintaining food safety and quality. By leveraging precise thermal control technologies, engineers can create systems that not only keep food safe but also enhance energy efficiency, reducing the overall environmental impact.
What role does freezing point play in climate science?
The freezing point of water at 32°F is essential in climate science as it directly influences weather patterns and climatic conditions. For instance, the freezing and thawing cycles of water bodies can impact local ecosystems and global climate systems, affecting everything from water cycle dynamics to regional temperature variations. These changes are pivotal for understanding and predicting weather patterns and climate changes.
How does cryogenic technology use freezing points?
Cryogenic technology uses the principles of freezing points extensively to store biological materials at temperatures close to 32°F. By maintaining materials at these low temperatures, scientists can preserve the structure and function of biological tissues, cells, and even entire embryos for extended periods. This is vital in medical research, allowing breakthroughs in fields such as regenerative medicine and genetic research.
As we delve deeper into the intricacies of temperature regulation, it becomes clear that understanding the freezing point at 32°F goes beyond academic curiosity; it has profound practical implications in both natural and engineered systems. From influencing ecosystems and weather patterns to revolutionizing medical science and enhancing food preservation methods, the science unveiled at this temperature point truly shapes our world in tangible and meaningful ways.