Gas dissolved in liquid is an area of study that is pivotal in various scientific and industrial contexts, from understanding biological processes to optimizing chemical reactions. The concept, while seemingly straightforward, encompasses a myriad of principles rooted in thermodynamics, physical chemistry, and chemical engineering. This article aims to delve into the intricacies of gas-liquid interactions, offering a deep dive into key insights and practical applications.
The Fundamentals of Gas Solubility in Liquids
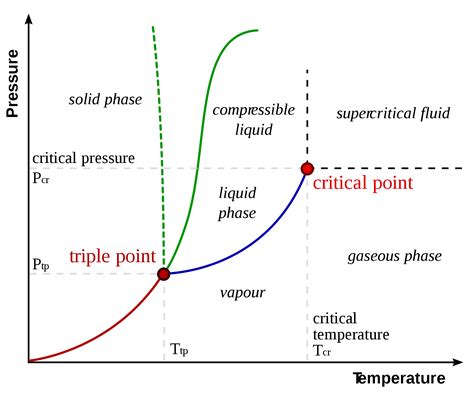
The solubility of gas in liquid is primarily governed by Henry’s Law, which states that the amount of gas that dissolves in a liquid is directly proportional to the partial pressure of that gas above the liquid. This fundamental relationship is expressed as:
C = k * P
where C represents the concentration of the dissolved gas, k is the Henry’s law constant, and P is the partial pressure of the gas. This principle is critical in understanding processes like carbonation in beverages, where the pressure of CO2 above the liquid determines the concentration of dissolved CO2.
Real-World Application: Carbonation in Beverages
A quintessential example is the carbonation process in soft drinks. During the carbonation process, carbon dioxide (CO2) is pressurized into the liquid under high pressure. As the container is opened, the pressure drops, leading to a release of CO2 in the form of bubbles—this is an excellent demonstration of Henry’s Law in action. The solubility of CO2 in water decreases as the pressure decreases, resulting in the familiar fizz.
Key Insights
- Primary insight: The solubility of gas in liquid is directly influenced by the partial pressure of the gas, a principle encapsulated in Henry's Law.
- Technical consideration: Temperature and the presence of other solutes can affect gas solubility, making it a multi-faceted phenomenon.
- Actionable recommendation: For industries relying on gas solubility, monitoring and controlling pressure and temperature conditions are critical.
Impact of Temperature and Pressure
Gas solubility in liquids is also profoundly affected by temperature. Generally, an increase in temperature leads to a decrease in gas solubility in liquid—a principle that underscores the process of degassing. When heated, gases have more kinetic energy and are less likely to remain dissolved in the liquid, thus tending to escape. This is why carbonated beverages taste flat after being left out at room temperature for too long: the CO2 is escaping due to the lower temperature and pressure conditions.
Additionally, pressure plays a crucial role in gas solubility. Higher pressures generally increase the amount of gas that can be dissolved in a liquid. This principle is exploited in deep-sea diving where divers are exposed to compressed air, altering the solubility of gases like nitrogen in the bloodstream.
Engineering Implications: Reactor Design and Optimization
In chemical engineering, understanding gas solubility in liquids is essential for reactor design and process optimization. In a gas-liquid reactor, efficient mixing and mass transfer are critical for achieving desired reaction rates. The design must account for the solubility characteristics of reactants to ensure optimal reaction conditions. For instance, in the production of nitric acid, the solubility of nitrogen oxides in water must be maximized to enhance the overall yield and efficiency of the process.
Moreover, this knowledge aids in the design of biological reactors such as fermenters used in the production of biofuels and pharmaceuticals. In these systems, oxygen solubility in water is a limiting factor for aerobic microorganisms. Reactors are engineered to maximize oxygen transfer rates, ensuring that microorganisms receive the necessary oxygen to thrive and produce the desired products.
FAQ Section
How does temperature affect gas solubility in liquids?
Generally, an increase in temperature decreases gas solubility in liquids. This is because gases have more kinetic energy at higher temperatures, making them less likely to remain dissolved in the liquid.
What role does pressure play in gas solubility?
Higher pressures generally increase the solubility of gases in liquids. This principle is utilized in industries such as carbonated beverages, where increased pressure enhances the solubility of carbon dioxide in water.
Understanding gas solubility in liquids is a critical aspect of numerous scientific and industrial processes. From everyday applications like carbonation to complex engineering processes in reactor design, the principles of gas-liquid interactions are indispensable. By mastering these fundamental concepts, professionals can optimize systems and processes, enhancing efficiency and productivity across various sectors.