Understanding the conversion between grams and liters is fundamental for many practical applications, ranging from cooking and baking to chemistry and science projects. This guide dives deep into the conversion process, offering step-by-step guidance, real-world examples, and practical solutions to ensure you have all the knowledge you need for precise measurements. This guide aims to demystify the process, addressing common user pain points and providing actionable advice to help you make accurate conversions with ease.
Quick Reference
Quick Reference
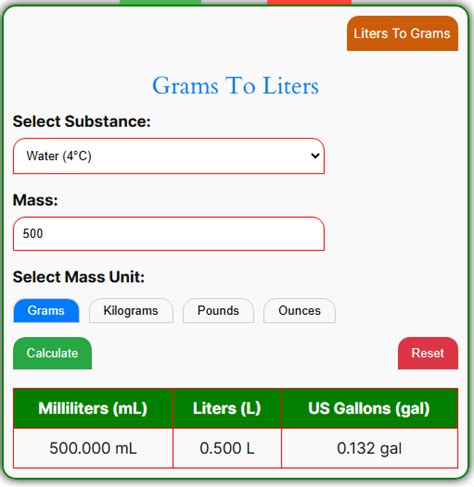
- Immediate action item with clear benefit: Use water as a default for gram to liter conversion. 1 gram of water = 0.001 liters.
- Essential tip with step-by-step guidance: To convert grams to liters, divide the weight in grams by the density of the substance in grams per liter.
- Common mistake to avoid with solution: Confusing weight and volume. Remember, density varies by substance, so always check the specific density for accurate conversion.
Converting Grams to Liters: The Basics
When dealing with the conversion from grams to liters, the first thing to understand is that this conversion isn’t straightforward for all substances. The key to making this conversion lies in the substance’s density. Here’s a step-by-step guide to understand and apply the conversion:
- Identify the Substance: Determine the substance you are measuring. The density of different materials varies significantly. For instance, the density of water is approximately 1 gram per milliliter (g/mL), while the density of olive oil is about 0.92 g/mL.
- Check Density: Find the density of the specific substance. This can often be found in scientific references, online databases, or through physical property tables. Density is typically expressed in grams per milliliter (g/mL) or grams per cubic centimeter (g/cm³).
- Conversion Formula: Use the formula: Liters = Grams / (Density in g/mL). Remember to convert milliliters to liters by dividing by 1000 since 1 liter equals 1000 milliliters.
Let’s apply this formula with an example using water:
- Example: If you have 2000 grams of water:
- First, identify that the density of water is 1 g/mL.
- Convert grams to liters: 2000 grams / (1 g/mL) = 2000 mL.
- Convert milliliters to liters: 2000 mL / 1000 = 2 liters.
Practical Examples of Gram to Liter Conversion
Let’s dive deeper with practical examples that cover various substances to illustrate the conversion process effectively:
Example 1: Converting Grams of Oil to Liters
Suppose you want to convert 1500 grams of olive oil to liters. The density of olive oil is approximately 0.92 g/mL.
- Identify the substance: olive oil.
- Check density: 0.92 g/mL.
- Apply the formula: Liters = Grams / Density. 1500 grams / 0.92 g/mL = 1630.43 mL.
- Convert milliliters to liters: 1630.43 mL / 1000 = 1.63 liters.
Example 2: Converting Grams of Honey to Liters
To convert 2000 grams of honey to liters, knowing that honey has a density of about 1.45 g/mL:
- Identify the substance: honey.
- Check density: 1.45 g/mL.
- Apply the formula: Liters = Grams / Density. 2000 grams / 1.45 g/mL = 1379.31 mL.
- Convert milliliters to liters: 1379.31 mL / 1000 = 1.38 liters.
Advanced Conversion Techniques
For more complex scenarios, such as dealing with substances with different densities or mixtures, you need to approach the conversion with additional precision:
- Substances with Variable Density: For substances like sugar solutions or saline solutions, the density can vary depending on concentration. Always ensure you have the accurate density for the specific concentration you are measuring.
- Mixtures: When converting substances within mixtures, calculate the density of the mixture if you know the proportions of each component. This might involve averaging the densities of the components or using a weighted average if the proportions are not equal.
- Advanced Equipment: For precision in labs, using equipment like a pycnometer or hydrometer can provide more accurate density measurements, leading to more precise conversions.
Practical FAQ
How do I convert grams to liters for a gas?
Converting grams to liters for gases is more complex because gases expand to fill their container, and density varies with temperature and pressure. You need to apply the Ideal Gas Law, PV=nRT, where P is pressure, V is volume, n is the number of moles, R is the gas constant, and T is temperature in Kelvin. Convert grams to moles using the substance’s molar mass, then use the Ideal Gas Law to find volume. Note: For practical purposes, this is best done with specialized equipment or software.
Why does the density of a substance affect the conversion?
Density is mass per unit volume. Since volume and mass are inversely related for a given substance, knowing the density is crucial for converting grams to liters. The higher the density, the fewer liters you’ll get from a certain number of grams.
How can I ensure accurate density values?
For precise conversions, always refer to reliable sources for density values. For substances with changing densities, such as solutions or mixtures, measure the density directly if possible. For everyday applications, trusted scientific databases and reference books are good sources.
This comprehensive guide should equip you with the knowledge to confidently convert grams to liters for a variety of substances, addressing the challenges users commonly face with precise measurements.