Halogens Periodic Table: Uncover Their Secrets and Impact
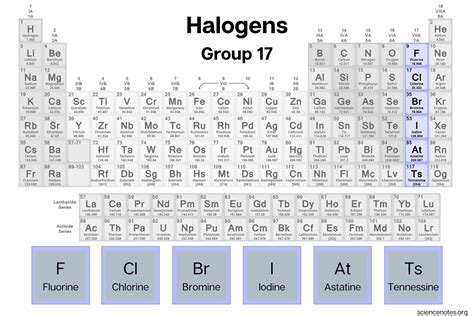
The halogens on the periodic table are a fascinating group of elements that include fluorine, chlorine, bromine, iodine, and astatine. They are known for their reactivity and significant impact on both industrial processes and everyday life. This guide will provide you with a step-by-step, practical approach to understanding the secrets of the halogens and how to leverage their properties effectively. Let’s dive right in and explore the unique characteristics and uses of these reactive elements.
Understanding the Problem: The Reactive Mysteries of Halogens
Halogens are highly reactive nonmetals, which makes them intriguing but challenging to handle. Their reactivity is due to their high electronegativity and the tendency to gain one electron to complete their outer electron shell, forming -1 ions. This characteristic leads to a range of practical applications, from disinfectants to flame retardants, but also poses challenges such as handling and storage. Many users struggle with understanding how to use halogens safely and effectively in various applications. This guide aims to demystify these elements and provide actionable advice to unlock their potential.
Quick Reference
Quick Reference
- Immediate action item: When working with halogens, always wear protective gear such as gloves and goggles to prevent any direct contact.
- Essential tip: Learn the reactivity order to predict the outcomes of halogen interactions, with fluorine being the most reactive and astatine the least.
- Common mistake to avoid: Mishandling halogens can lead to dangerous reactions; always store them in proper containers away from incompatible materials.
Essential Basics: Getting Started with Halogens
To get started with halogens, it’s essential to understand their basic properties and common uses. This section will cover the fundamental aspects of each halogen, from their atomic structure to their primary applications.
Fluorine (F) is the lightest halogen and the most reactive, making it crucial in various industries, from manufacturing fluorocarbons to dental products. Chlorine (Cl) is widely used for its disinfecting properties in water treatment and manufacturing plastics like PVC. Bromine (Br) finds use in flame retardants, drilling fluids, and petrochemical industries. Iodine (I) is vital for medical applications such as disinfectants and thyroid treatment, while astatine (At) is extremely rare and mostly studied for its radioactive isotopes.
Detailed How-To: Handling Fluorine
Fluorine is the most reactive of the halogens and poses significant handling risks. Below, you’ll find detailed steps and best practices for safely working with fluorine.
- Safety Gear: Always wear full protective clothing including a face shield, chemically resistant gloves, and safety goggles when handling fluorine. Never use plastic containers due to its reactive nature.
- Storage: Store fluorine in a specially designed, inert container made of copper or another non-reactive metal. Keep it in a secure, well-ventilated area away from any organic material and oxidizing agents.
- Usage: Fluorine is used in the production of fluorocarbons, which are essential in many chemical industries. It is also used in the synthesis of various compounds, including uranium hexafluoride for nuclear reactors.
Remember, fluorine reacts violently with water and organic materials. Proper training and adherence to safety protocols are critical to prevent accidents.
Detailed How-To: Utilizing Chlorine
Chlorine’s disinfecting properties make it invaluable for water treatment and sanitation. Here’s how to use chlorine safely and effectively:
- Application in Water Treatment: Chlorine is commonly added to public water supplies to kill bacteria and viruses. It’s also used in swimming pools to maintain clean and safe water. Always follow manufacturer guidelines for dosage to avoid over-chlorination, which can lead to harmful byproducts.
- Safety Measures: When handling chlorine gas, use a gas mask and ventilated area. For liquid chlorine, wear protective gloves and clothing. Ensure you have appropriate spill kits and neutralizing agents like sodium bisulfite handy in case of accidental releases.
- Storage: Store chlorine gas in steel containers equipped with appropriate pressure relief devices. Keep them away from heat sources and incompatible materials like oil or grease.
Chlorine is also used in the production of polyvinyl chloride (PVC) and other chlorinated compounds. It’s essential to follow all safety guidelines to handle it effectively.
Detailed How-To: Employing Bromine
Bromine’s unique properties make it suitable for various industrial applications, from flame retardants to drilling fluids. Here’s how to utilize bromine safely:
- Usage in Flame Retardants: Bromine is a key component in many flame retardant chemicals. Ensure proper ventilation when using these products and follow all environmental and safety regulations.
- Handling Precautions: Bromine can cause skin and respiratory irritation. Wear full-body protective clothing, gloves, and a respirator when handling. Ensure workspaces are well-ventilated.
- Storage: Store bromine in tightly sealed containers made of compatible materials such as glass or stainless steel. Keep them in cool, well-ventilated areas away from organic materials and heat sources.
Bromine is also used in the petrochemical industry, and its handling requires strict adherence to safety protocols to mitigate health and environmental risks.
Detailed How-To: Applying Iodine
Iodine’s critical role in medical and health-related applications makes it a valuable element. Here’s how to apply iodine effectively:
- Medical Uses: Iodine is used in antiseptic solutions like Betadine, which are essential in preventing infections during medical procedures. Always follow the manufacturer’s instructions for application and dilution.
- Nutrient Supplementation: Iodine is a vital nutrient required for thyroid function. Supplements are available, but it’s essential to consult a healthcare provider to determine the correct dosage.
- Safety Practices: Iodine can be corrosive and cause skin irritation. Use protective gloves and clothing when handling. Store iodine solutions in dark, airtight containers to prevent decomposition.
In industrial applications, iodine is used in organic synthesis and photographic chemicals, necessitating careful handling and storage to prevent exposure.
Practical FAQ
Can halogens be safely mixed with other elements?
Halogens should not be mixed with incompatible elements without thorough knowledge of their chemical properties and potential reactions. For example, mixing fluorine with organic materials can be highly explosive. Always consult the Material Safety Data Sheet (MSDS) and follow guidelines for safe mixing and storage of halogens.
What should I do in case of a halogen spill?
In the event of a halogen spill, immediately evacuate the area and call for professional assistance. Follow these steps for immediate action:
- Do not touch the spill directly; leave it to trained professionals.
- Evacuate the area and close all doors to prevent inhalation.
- Use appropriate spill containment kits and neutralizing agents specific to the halogen involved.
How do I dispose of halogen-containing waste safely?
Disposal of halogen-containing waste must adhere to local, state, and federal regulations. Here’s a step-by-step guide:
- Consolidate the waste in labeled, secure containers.
- Contact a certified hazardous waste disposal company.
- Follow all instructions provided by the disposal service to ensure safe handling and transportation.
Advanced Techniques: Leveraging Halogens in Industry
Once you’re comfortable with the basics of handling