Understanding the molecular geometry of hydrogen cyanide (HCN) is a fundamental aspect of chemistry that provides insight into its structure, bonding, and reactivity. This guide will walk you through the essential steps to grasp HCN’s molecular geometry, using practical examples and actionable advice to enhance your understanding. Whether you’re a student tackling a new topic or a professional looking to refresh your knowledge, this guide aims to address your needs directly.
Why Understanding HCN Geometry Matters
Hydrogen cyanide, HCN, is a simple yet highly intriguing molecule. Its linear geometry is crucial for its unique chemical properties, making it a vital compound in various industrial applications and organic synthesis. Understanding its molecular geometry helps predict its behavior in reactions, its physical properties, and its environmental impact. This guide will demystify HCN’s structure, focusing on practical applications and real-world examples to make your learning experience both comprehensive and enjoyable.
Getting Started: The Basics of Molecular Geometry
Before diving into HCN’s specific geometry, it’s important to grasp the fundamentals of molecular geometry in general. Molecular geometry refers to the three-dimensional arrangement of atoms in a molecule and is primarily determined by the repulsion between electron pairs around a central atom. The key to understanding molecular geometry lies in the principles of VSEPR (Valence Shell Electron Pair Repulsion) theory, which predicts how electron pairs around a central atom will arrange themselves to minimize repulsion.
Quick Reference
Quick Reference
- Immediate action item: Review VSEPR theory basics to understand electron pair repulsion.
- Essential tip: Count the number of valence electron pairs around the central atom to predict molecular shape.
- Common mistake to avoid: Mistaking the molecular shape with the electron pair geometry; they are different concepts.
Detailed How-To Guide on HCN Molecular Geometry
Now that you’ve revisited the basics of molecular geometry, let’s delve into the specifics of HCN. HCN is a diatomic molecule consisting of one hydrogen atom and one carbon atom bonded to a nitrogen atom. Despite its simplicity, its linear shape is derived from a unique electronic configuration and minimal repulsion due to having only two atoms.
Step-by-Step to HCN Geometry
To determine the geometry of HCN, follow these steps:
Step 1: Identify the Central Atom
In HCN, carbon © is the central atom. Carbon lies between hydrogen (H) and nitrogen (N). This placement ensures that carbon’s electron pairs are positioned to minimize repulsion.
Step 2: Count Valence Electrons and Electron Pairs
Carbon has four valence electrons, nitrogen has five, and hydrogen has one. Together, they form a total of 10 valence electrons in HCN. These electrons are shared between carbon and nitrogen, and between nitrogen and hydrogen, respectively.
To determine the electron pairs, divide the valence electrons by two: 10/2 = 5 electron pairs. Since each bond counts as one electron pair, we have two bonds: one between C and N, and another between N and H.
Step 3: Apply VSEPR Theory
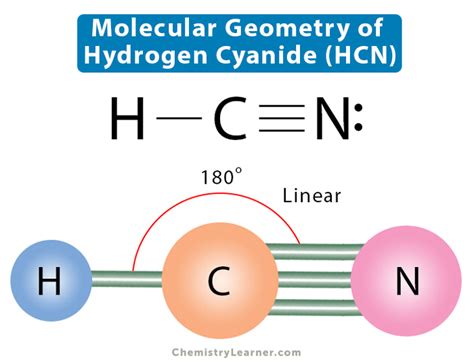
According to VSEPR theory, electron pairs will arrange themselves to minimize repulsion. For HCN, there are two bonding pairs and no lone pairs on the central atom. This configuration leads to a linear geometry where the angle between the atoms is 180 degrees.
The linear shape results from the fact that the two electron pairs are directly opposite each other, which minimizes repulsion and creates the most stable configuration for the molecule.
Step 4: Visualizing the Geometry
To visualize HCN’s geometry, imagine a straight line where the carbon atom is in the middle, bonded to a nitrogen atom on one side and a hydrogen atom on the other side. This linear structure is crucial because it influences HCN’s polarity and its ability to participate in specific types of chemical reactions.
Advanced Considerations in HCN Geometry
As you become more comfortable with the basics, consider these advanced aspects of HCN geometry:
Molecular Orbital Theory
Beyond VSEPR theory, understanding molecular orbital theory can offer deeper insights into the bonding in HCN. The σ (sigma) bond between carbon and nitrogen involves the overlap of sp hybridized orbitals from carbon and a p orbital from nitrogen, creating a stable and linear molecule.
Resonance Structures
Although HCN primarily exhibits a single Lewis structure, discussing resonance can help understand the stability and partial charges in molecules with double or triple bonds. For HCN, the single bond structure is most stable, but understanding resonance principles can deepen your comprehension of molecular stability.
Electronegativity and Polarity
The difference in electronegativity between carbon, nitrogen, and hydrogen contributes to the polarity of the HCN molecule. Nitrogen is more electronegative than carbon, and hydrogen is least electronegative, resulting in a polar molecule with a dipole moment along the bond axis.
Practical FAQ
How does HCN’s geometry affect its reactivity?
HCN’s linear geometry directly impacts its reactivity. Being a polar molecule due to its linear shape and electronegativity differences, HCN can act as a nucleophile in reactions where it donates its lone pairs to electrophiles. This reactivity is also influenced by the position of the atoms, as the central carbon atom’s sigma bond orientation facilitates specific types of interactions, especially in organic synthesis.
Can HCN’s geometry change under different conditions?
In most conditions, HCN maintains its linear geometry because the central carbon’s sigma bonds with nitrogen and hydrogen create a stable configuration that resists change. However, under extreme conditions such as high pressure or in complex chemical reactions, other structural configurations might form temporarily, but these are exceptions rather than the rule.
Tips and Best Practices
To master HCN molecular geometry, consider these tips and best practices:
- Practice drawing Lewis structures for different molecules to understand the electron pair distribution and how it influences molecular shape.
- Use molecular modeling kits or software to visualize molecular geometries and better grasp spatial arrangements.
- Regularly review VSEPR theory principles and apply them to a wide range of molecules, not just HCN.
- Engage with molecular geometry through real-world examples, like understanding how HCN is used in industrial processes such as the production of dyes and agrochemicals.
By following this guide, you’ll gain a thorough understanding of HCN’s molecular geometry, equipping you with the knowledge to explore its applications and implications in chemistry and beyond.