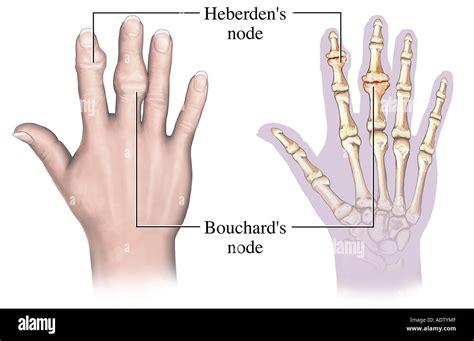
Heberden's and Bouchard's nodes are commonly encountered manifestations of osteoarthritis, primarily affecting the finger joints. For those navigating the labyrinth of degenerative joint conditions, understanding these nodes involves delving into their origins, clinical presentations, and current treatment strategies. These anatomical nodules, often dismissed as mere signs of aging, are actually indicative of complex biochemical and biomechanical processes. Their progression, impact on function, and therapeutic management form the cornerstone of rheumatologic and orthopedic discourse. The quest to manage these conditions effectively requires not only a grasp of their pathophysiology but also an appreciation of the nuanced options available within modern medicine. As a specialist with decades of clinical experience and a research background in degenerative joint diseases, I aim to distill the latest insights into Heberden's and Bouchard's nodes, offering clarity and evidence-based guidance to both clinicians and informed patients alike.
Key Points
- Heberden's and Bouchard's nodes are physical signs of osteoarthritis, representing osteophyte formation at distal and proximal interphalangeal joints respectively.
- The primary causes involve mechanical stress, genetic predisposition, and biochemical alterations leading to cartilage degradation.
- Symptoms often include joint swelling, stiffness, and pain, which can impair fine motor skills and daily functioning.
- Treatment approaches range from conservative management, including pharmacotherapy and physical therapy, to surgical intervention in advanced cases.
- Emerging biological therapies and lifestyle modifications show promise in slowing disease progression and alleviating symptoms.
Pathophysiology and Etiology of Heberden’s and Bouchard’s Nodes
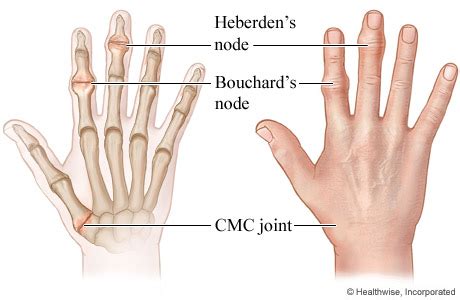
The formation of Heberden’s and Bouchard’s nodes reflects a cascade of degenerative processes intrinsic to osteoarthritis (OA). The key pathological event begins with the breakdown of articular cartilage in the interphalangeal joints, instigated by an imbalance between anabolic and catabolic factors within cartilage matrix homeostasis. Mechanical loading plays a critical role; repetitive stress, joint overuse, and microtrauma exacerbate cartilage wear, particularly in individuals with genetic susceptibility or joint malalignment.
osteophyte formation — the hallmark of these nodes — results from an attempted repair process. Osteoblasts proliferate at joint margins, depositing new bone in areas of cartilage loss. These bony outgrowths tend to encroach on joint space, causing palpable nodules. Histologically, these nodules contain fibrocartilaginous tissue, hypertrophic chondrocytes, and mineralized deposits, indicating ongoing remodeling and degeneration processes.
Genetic studies have shed light on familial patterns, pointing to polymorphisms in genes regulating cartilage extracellular matrix components and inflammation, such as COL2A1 and IL-1β. Hormonal factors, particularly estrogen deficiency in postmenopausal women, also influence disease prevalence and severity, aligning with epidemiologic data that demonstrate a higher incidence in females after menopause.
Biomechanical Contributors and Risk Factors
Beyond genetic predisposition, biomechanical influences underpin the development of these nodal osteophytes. Repetitive gripping, heavy manual labor, or occupations necessitating fine motor control have all been implicated as risk factors. Such activities increase load transmission across finger joints, accelerating cartilage deterioration and osteophyte proliferation. Additionally, joint misalignments, such as acute deviations or ligamentous laxity, impose uneven stress distribution, fostering localized degenerative changes.
| Relevant Category | Substantive Data |
|---|---|
| Prevalence in Population | Estimated at 10-20% in adults over 50, with higher rates in women (up to 75%) |
| Genetic Influence | Familial aggregation observed in approximately 50% of cases |
| Progression Rate | Worsening of nodal size and joint function over 5-10 years in most patients |
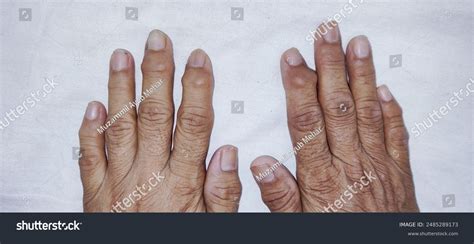
Clinical Manifestations and Diagnostic Approach
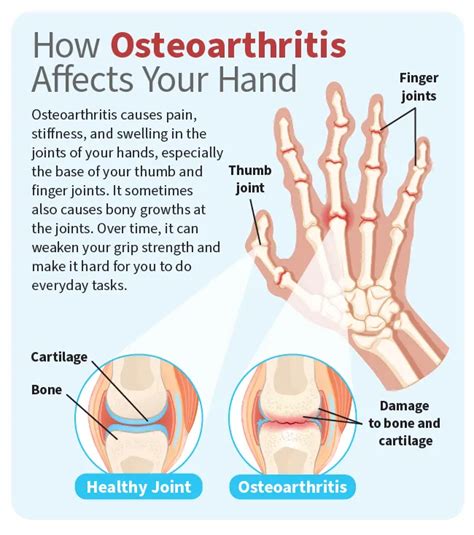
The presence of Heberden’s and Bouchard’s nodes is often a tip-off for clinicians to investigate underlying osteoarthritic changes. Patients typically report joint stiffness, especially after periods of inactivity, accompanied by mild to moderate pain localized over the joint margins. Visible swelling and palpable nodules are characteristic findings, with Heberden’s nodes appearing at the distal interphalangeal (DIP) joints and Bouchard’s at the proximal interphalangeal (PIP) joints.
Physical examination reveals these bony outgrowths, which can vary from soft and pliable in early stages to hard and immobile as sclerosis progresses. Reduced range of motion and deformities such as boutonnière or, less commonly, swan-neck deformities may develop over time. Notably, while these nodes signify OA, they are also valuable diagnostic markers that, in conjunction with radiographic evidence, aid in confirming the diagnosis.
Imaging and Laboratory Investigations
Radiography remains the gold standard for assessing osteophyte size, joint space narrowing, subchondral sclerosis, and cyst formation. Typical radiographic features include marginal osteophytes at joint margins and joint space reduction, often accompanied by subchondral sclerosis.
Advanced imaging, like MRI, can provide detailed insights into cartilage integrity, synovial inflammation, and early subchondral changes, but is generally reserved for complex cases. Laboratory tests are usually unremarkable, as osteoarthritis is primarily a degenerative rather than an inflammatory process, although markers like serum hyaluronic acid and Cartilage Oligomeric Matrix Protein (COMP) can reflect cartilage turnover and may assist in disease monitoring.
Conventional and Emerging Treatment Strategies
Managing Heberden’s and Bouchard’s nodes necessitates a multimodal approach focusing on alleviating symptoms, preserving joint function, and slowing disease progression. Current guidelines endorse conservative measures as first-line, emphasizing pharmacological, physical, and lifestyle interventions. In refractory cases, surgical options can restore joint integrity and function.
Pharmacotherapy and Physical Management
Nonsteroidal anti-inflammatory drugs (NSAIDs) remain the cornerstone for symptomatic relief, with topical formulations like diclofenac gel offering localized benefit and fewer systemic effects. Acetaminophen, though less potent, may be considered in patients with contraindications to NSAIDs. Additionally, symptomatic improvement can be achieved with corticosteroid injections into affected joints, although repeated use is limited due to potential cartilage toxicity.
Physical therapy, including range-of-motion exercises, joint protection techniques, and splinting, helps maintain mobility and reduce deformity progression. Patient education on ergonomic modifications and activity adjustments forms a vital component of management, preventing exacerbation of degenerative changes.
Biological and Lifestyle Interventions
Emerging therapies targeting inflammatory mediators, such as biologic agents that inhibit interleukin-1 or tumor necrosis factor-alpha, are under investigation, with some showing promising effects in slowing cartilage degradation. Lifestyle modifications — weight management, balanced nutrition rich in omega-3 fatty acids, and regular low-impact exercise — are associated with reduced symptom severity and better joint longevity.
| Relevant Category | Substantive Data |
|---|---|
| NSAID Efficacy | Significant symptom reduction in 60-70% of patients, but long-term use linked to gastrointestinal and cardiovascular risks |
| Exercise Benefits | Improves joint function in 80% of participants, with reduced pain and stiffness |
| Surgical Interventions | arthroplasty or joint fusion indicated in severe cases; success rates >85% |
Surgical and Minimally Invasive Options
When conservative measures fail, surgical correction offers definitive treatment. Procedures include joint debridement, osteophyte removal, or even arthroplasty, aimed at restoring function and dismantling symptomatic nodules. The decision hinges on disease severity, functional impairment, and patient preference.
Minimally invasive techniques such as arthroscopic removal of osteophytes are gaining traction due to reduced recovery times and lower complication rates. Nonetheless, the risk of recurrence and postoperative joint instability must be critically weighed.
Postoperative Rehabilitation and Outcomes
Rehabilitation protocols emphasize early mobilization, physiotherapy, and patient education to optimize functional recovery. Long-term, many patients report significant pain relief and improved dexterity, affirming surgery’s role as a valuable option in advanced disease stages.
What differentiates Heberden’s from Bouchard’s nodes in clinical practice?
+Heberden’s nodes are located at the distal interphalangeal (DIP) joints, whereas Bouchard’s nodes appear at the proximal interphalangeal (PIP) joints. Clinically, Heberden’s are often more prominent and can cause visible deformity, while Bouchard’s may be less noticeable but contribute to similar functional limitations.
Are there lifestyle modifications that effectively slow the progression of these nodal osteoarthritis?
+Yes, engaging in low-impact activities like swimming or tai chi, maintaining a healthy weight, using ergonomic tools, and avoiding repetitive stress can help reduce joint load. These strategies aim to minimize mechanical stress and inflammation, potentially slowing disease progression.
Can early intervention prevent the development of prominent nodules?
+While complete prevention isn’t always possible due to genetic and age-related factors, early diagnosis and proactive management — including activity modification, joint protection, and pharmacologic therapy — can mitigate severity and delay nodule formation.