Unveiling Nitric Acid’s Power: Understanding HNO3’s Strength
Are you puzzled by whether nitric acid (HNO3) is strong or weak, or how its power can be harnessed in practical applications? You’re not alone. Nitric acid is one of the most widely used acids in both laboratory and industrial settings due to its potent oxidizing properties. In this guide, we’ll decode the chemistry behind nitric acid, explore its strengths, provide practical examples of its applications, and address common concerns. By the end, you’ll have a clear understanding of how to safely and effectively use this powerful acid.
Immediate Action: Safety First
Before diving into the specifics of HNO3’s strength, it’s crucial to understand the safety aspects. Nitric acid is highly corrosive and can cause severe burns upon contact with skin. Always handle it with appropriate personal protective equipment (PPE) including gloves, goggles, and a lab coat. Use it in well-ventilated areas to avoid inhaling fumes. An immediate action item is to ensure you have a spill kit and eyewash station nearby in case of accidental exposure.
Quick Reference
- Immediate action item: Always wear appropriate PPE and work in a well-ventilated area.
- Essential tip: Keep a spill kit and eyewash station accessible.
- Common mistake to avoid: Not wearing PPE or working in a poorly ventilated area.
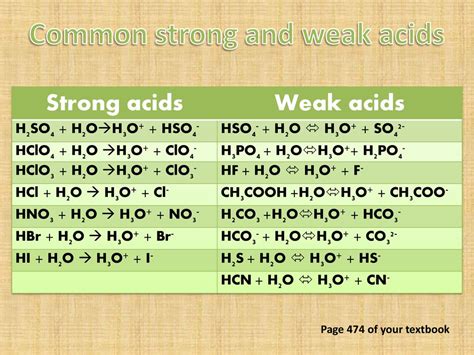
Is HNO3 a Strong Acid?
Nitric acid (HNO3) is classified as a strong acid. This classification is due to its high degree of ionization in water, where it almost completely dissociates into hydrogen ions (H+) and nitrate ions (NO3-). This strong tendency to donate protons makes it incredibly effective in various chemical reactions, especially in processes that require a strong acid.
Real-World Example: In the manufacturing of fertilizers, nitric acid is used to produce nitrates. The strong acid properties ensure efficient conversion of raw materials into desired compounds. This makes it indispensable in the agrochemical industry.
Detailed How-To: Preparing a Nitric Acid Solution
To make a nitric acid solution, follow these detailed steps to ensure accuracy and safety:
- Step 1: Gather your materials. You’ll need nitric acid (HNO3), a container with a lid, PPE (gloves, goggles, lab coat), and a spill kit.
- Step 2: Ensure you are in a well-ventilated area or a fume hood. Open the container carefully to avoid splashing. Pour the nitric acid slowly into the water, not vice versa, to prevent a violent reaction.
- Step 3: Stir the solution gently with a glass rod or a stirrer to help the acid dissolve completely. Do not use a metal stirrer as it may react with the acid.
- Step 4: Once dissolved, cover the container with a lid and label it appropriately with the concentration and date of preparation. Store it in a cool, dark place away from incompatible materials.
These steps are fundamental for preparing nitric acid safely and accurately. Each action ensures the integrity of the solution and your safety in the process.
Detailed How-To: Using Nitric Acid in Reactions
When using nitric acid in chemical reactions, it’s important to understand its oxidizing properties. This makes it highly effective in reactions where oxidation is desired:
- Step 1: Identify the reaction requirements. Nitric acid is often used in oxidation reactions, particularly in the preparation of nitro compounds.
- Step 2: Add the acid to your reaction mixture slowly. The exothermic reaction can be vigorous, so adding it slowly helps control the heat generated.
- Step 3: Monitor the temperature. The reaction heat can rise quickly, so use a temperature-controlling device if necessary.
- Step 4: Stir continuously. This helps in maintaining homogeneity and ensures the reaction proceeds evenly.
- Step 5: After the reaction is complete, neutralize any remaining acid with a weak base like sodium bicarbonate (NaHCO3). Always follow with water to ensure complete neutralization.
By following these steps, you can harness nitric acid’s powerful oxidizing properties effectively and safely.
FAQs: Common User Questions and Practical Answers
Can HNO3 be diluted to make it weaker?
Yes, nitric acid can be diluted with water to reduce its strength. Dilution lowers the concentration of hydrogen ions, thereby reducing its acidity. However, it’s crucial to remember that even diluted nitric acid remains hazardous. Always handle it with care and use appropriate safety measures.
What happens if I mix HNO3 with other acids?
Mixing nitric acid with other acids can lead to unpredictable reactions. It’s generally best to avoid mixing acids unless you have specific knowledge about the reaction outcomes. If a reaction requires multiple acids, they should be added separately and under controlled conditions. Always consult a chemical safety guide or expert before combining acids.
How do I dispose of leftover nitric acid?
Disposing of nitric acid requires following local regulations. Typically, it must be neutralized first using a suitable base before disposal. Neutralized acid should be diluted with water and poured into a designated hazardous waste container. Never pour acid down the drain. Follow all local, state, and federal regulations for hazardous waste disposal.
This guide provides comprehensive, actionable advice on understanding and using nitric acid safely and effectively. With these steps and tips, you can leverage HNO3’s powerful properties in your chemical endeavors while prioritizing safety and environmental responsibility.