Lithium’s Electron Count: Surprising Insight!
Welcome to an in-depth exploration of lithium's electron count, a topic that reveals fundamental scientific insights often overlooked in everyday discussions. Whether you are a curious student, a seasoned chemist, or simply someone fascinated by the invisible world of atoms, this guide will provide you with a comprehensive understanding of lithium's electron configuration. By breaking down the principles and providing practical examples, we aim to make complex scientific concepts accessible and easy to grasp.
Understanding the electron count of an element is crucial because it dictates the element's chemical properties, behavior in reactions, and interactions with other elements. In the case of lithium, knowing its electron count will give you a deeper appreciation for how it functions within the periodic table and in various applications, from batteries to medicine.
Why Electron Count Matters
An element’s electron count is more than just a number; it tells you about an element’s reactivity, how it bonds with other elements, and what kind of compounds it can form. Lithium, with its unique position in Group 1 of the periodic table, has an electron configuration that stands out due to its simplicity and significant impact on chemical behavior.
For many people, the idea of electrons swirling around a nucleus might seem abstract. However, when we delve into lithium, it becomes clear why its electron count—specifically, its arrangement—is pivotal in determining its role in everything from everyday technology to advanced scientific research.
Quick Reference
Quick Reference
- Immediate action item with clear benefit: Start by determining lithium’s atomic number (3) to immediately understand its electron count (3).
- Essential tip with step-by-step guidance: Learn to write lithium’s electron configuration: 1s2 2s1.
- Common mistake to avoid with solution: Don’t confuse electron count with protons or neutrons; they are distinct atomic components.
Understanding Lithium’s Electron Configuration
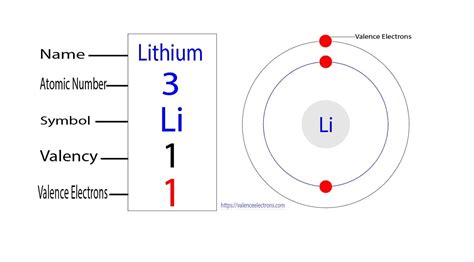
To understand lithium’s electron count, we need to start with some basics about atoms. An atom is composed of a nucleus containing protons and neutrons, surrounded by electrons. Each element has a unique number of protons, which defines it on the periodic table. Lithium’s atomic number is 3, meaning it has 3 protons and, in a neutral state, 3 electrons.
Now, let’s dive deeper into its electron configuration, which tells us how these electrons are distributed in the atom’s orbitals.
Step-by-Step Breakdown of Lithium’s Electron Configuration
Lithium’s electron configuration can be broken down into a few simple steps:
- Determine the number of electrons: Since lithium has an atomic number of 3, it has 3 electrons.
- Identify the electron shells: Electrons fill the lowest energy levels first. Lithium’s electrons fill the 1s and 2s orbitals.
- Fill the orbitals: The 1s orbital can hold up to 2 electrons, so the first two electrons fill this orbital (1s2). The third electron moves to the next available orbital, the 2s orbital (2s1).
- Write the configuration: Combine these to get lithium’s electron configuration: 1s2 2s1.
This configuration gives lithium its unique chemical properties, including its tendency to lose one electron easily, making it highly reactive.
Real-World Application of Lithium’s Electron Configuration
Understanding lithium’s electron count and configuration isn’t just academic. It has practical implications across various fields. For instance:
- In battery technology, lithium-ion batteries utilize lithium’s reactivity and electron count to store and release energy efficiently.
- In medicine, lithium salts are used to treat bipolar disorder, showing how this element’s chemical behavior can impact health directly.
- In scientific research, lithium’s simple electron configuration makes it an excellent model element for studying atomic behavior and reactions.
Practical Examples
To see how lithium’s electron configuration plays out in real scenarios, let’s look at two practical examples:
Example 1: Lithium in Batteries
Lithium-ion batteries are ubiquitous in modern technology, from smartphones to electric vehicles. The electron count of lithium is central to their function:
- Energy Storage: Lithium’s ability to donate electrons (oxidation) allows it to release energy, powering devices.
- Movement of Lithium Ions: During charging, lithium ions move into the anode (where they originate), and during discharge, they move to the cathode, providing the current.
This movement is a testament to lithium’s electron count and its role in electron transfer processes essential for battery functionality.
Example 2: Lithium in Medicine
In medicine, lithium’s chemical properties help treat mental health conditions:
- Bipolar Disorder Treatment: Lithium salts interfere with neurotransmitters and neurotransmission processes in the brain, balancing mood states and reducing episodes.
- Mechanism of Action: Lithium ions bind to certain enzymes and proteins in the brain, altering their activity and thus regulating mood.
Lithium’s chemical interactions, driven by its electron configuration, are crucial for its medical uses.
Common Questions and Answers
Why does lithium’s electron configuration matter in chemical reactions?
Lithium’s electron configuration—specifically its single electron in the 2s orbital—makes it highly reactive, especially in forming ionic bonds. This reactivity allows lithium to easily lose its outermost electron, becoming Li+ and forming compounds like LiCl (lithium chloride). Understanding this configuration helps predict lithium’s behavior in chemical reactions, aiding in fields like battery technology and pharmaceuticals.
How does lithium’s electron count affect its applications in technology?
Lithium’s unique electron count facilitates its use in high-capacity, lightweight batteries. Its ability to lose and gain electrons efficiently during charge and discharge cycles is key to the performance of lithium-ion batteries. This electron activity allows these batteries to deliver a high amount of energy with minimal weight, making them ideal for portable electronics and electric vehicles.
What are the safety considerations when working with lithium in a laboratory?
Lithium is highly reactive, especially with water, which can cause it to ignite. When working with lithium, it’s essential to:
- Handle lithium under inert atmospheres (like argon) to prevent exposure to moisture and air.
- Use proper personal protective equipment (PPE) such as gloves, goggles, and lab coats.
- Store lithium in airtight containers away from water and moisture.
Following these safety protocols ensures safe handling and prevents accidents related to lithium’s reactive nature.
By now, you should have a clearer picture of lithium’s electron count and its implications. This knowledge bridges the gap between basic chemistry and its vast applications, from tech gadgets to therapeutic medications. Understanding these principles not only enhances your scientific knowledge but also equips you with the insight to appreciate the practical importance of lithium in everyday life.
Remember, delving into the atomic structure and electron configurations of elements is more than just an academic exercise—it’s a journey into the very fabric of the universe, showing how the smallest particles play significant roles in our world.