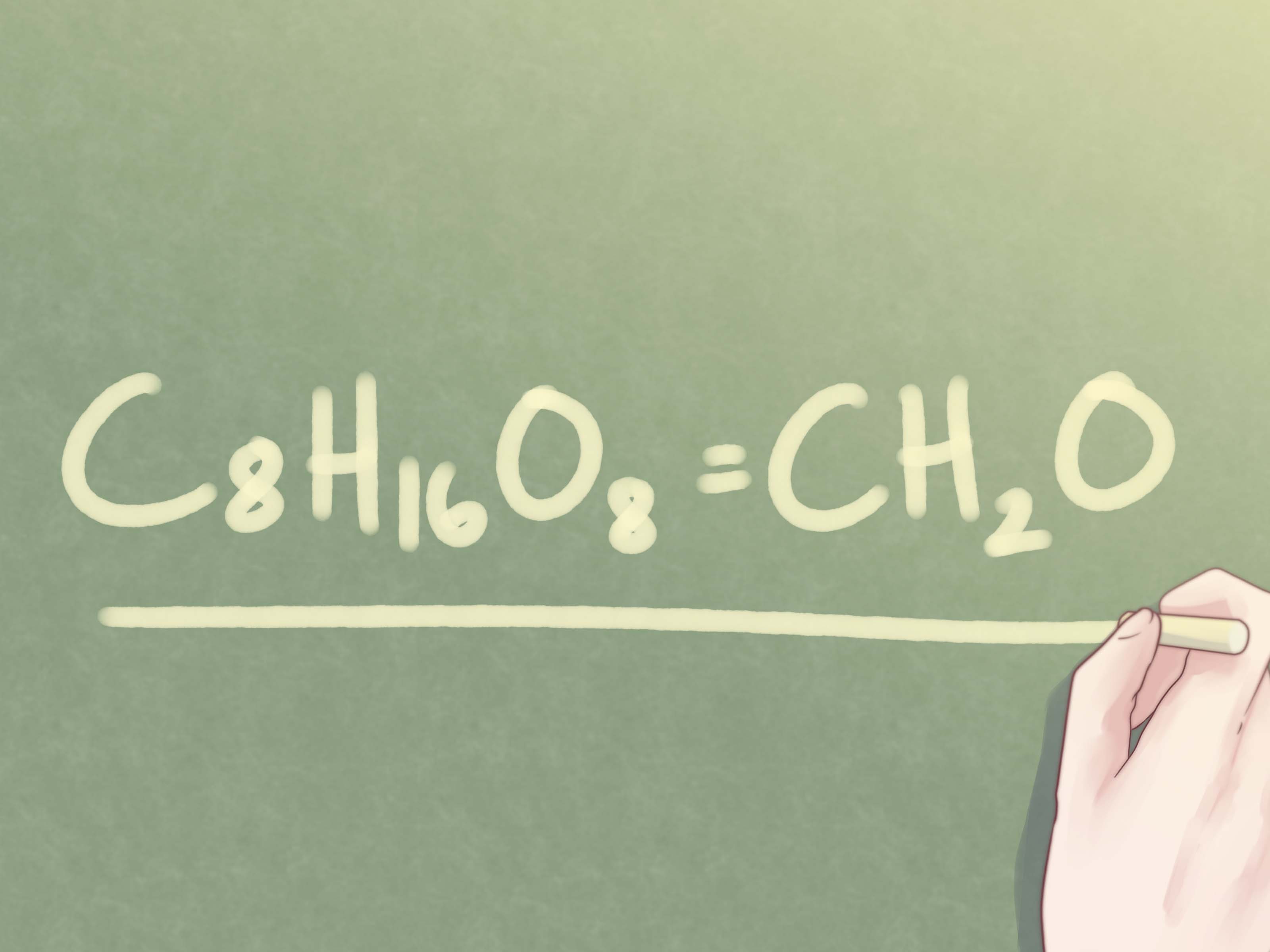If you’re feeling lost in the labyrinth of empirical formulas, you’re not alone. Many people struggle with the seemingly abstract concepts and calculations that involve figuring out the simplest whole-number ratio of atoms in a compound. This guide aims to decode empirical formulas by breaking down the process into practical, easy-to-follow steps. We’ll delve into real-world examples to illustrate the practical application of these formulas and offer actionable advice that even the most apprehensive novice can implement.
Understanding Empirical Formulas: The Key to Simplification
An empirical formula represents the simplest ratio of elements in a compound. Unlike molecular formulas, which show the exact number of atoms in a molecule, empirical formulas strip down the ratio to its most fundamental form. For example, when dealing with glucose, the molecular formula is C₆H₁₂O₆, but the empirical formula is CH₂O.
The importance of empirical formulas lies in their ability to give a quick and effective way of understanding the composition of substances, which is especially useful in fields like chemistry, biochemistry, and material science. Knowing how to decode these formulas is crucial for anyone looking to build a foundation in these disciplines.
Quick Reference
Quick Reference
- Immediate action item: Start with the compound’s molecular formula and identify the simplest ratio.
- Essential tip: Use the method of dividing by the greatest common divisor for integer ratios.
- Common mistake to avoid: Overcomplicating the problem by not simplifying the ratio to the smallest whole numbers.
Step-by-Step Guidance for Finding Empirical Formulas
Let’s dive into the step-by-step method for finding empirical formulas. This guide will take you through the process from the basics to more advanced techniques. We’ll break down complex steps into manageable parts so you can apply them easily.
Step 1: Understand the Molecular Formula
Before you start, ensure you have the molecular formula of the compound. This formula indicates the actual number of each type of atom in a molecule of the compound.
For example, let’s take ascorbic acid (Vitamin C), which has the molecular formula C₆H₈O₆.
Step 2: Convert Mole Ratios to Simplest Form
The goal is to convert the ratio of atoms into the simplest whole-number ratio. This process involves converting the molecular formula into an empirical formula.
Here’s how to do it:
- Calculate moles of each element: Start by finding the molar mass of each element in the molecular formula and then convert the given mass of each element into moles. For example, using Vitamin C (C₆H₈O₆):
- Carbon (C): 12 g/mol * 6 = 72 g/mol
- Hydrogen (H): 1 g/mol * 8 = 8 g/mol
- Oxygen (O): 16 g/mol * 6 = 96 g/mol
- Divide each value by the smallest value: To simplify the ratio, divide each mole value by the smallest number of moles calculated (in this case, 8).
- C: 72/8 = 9
- H: 8/8 = 1
- O: 96/8 = 12
- Use the resulting ratios: These ratios (9:1:12) represent the simplest whole number ratio of atoms.
Step 3: Simplify the Ratios if Necessary
Sometimes, the ratios may not be the simplest form. You may need to divide these numbers by their greatest common divisor (GCD) to get the simplest form.
For Vitamin C’s ratios (9:1:12), since they are already in their simplest form, we can write the empirical formula as C₉H₁O₁₂. However, to keep things simple, chemists often write it as CH₂O.
Step 4: Verification
Verify the simplicity by ensuring that all numbers are whole numbers and cannot be further simplified.
Practical Example: Finding the Empirical Formula of Sulfur and Oxygen Compound
Let’s take another example to illustrate this process. Suppose you have a compound with the molecular formula SO₂ but you want to find its empirical formula.
- Identify the molecular formula: SO₂ indicates 1 sulfur (S) atom and 2 oxygen (O) atoms.
- Calculate the molar mass of each element:
Element Atomic Mass S 32.07 g/mol O 16.00 g/mol (per atom) - Determine moles:
Element Molecular Mass Contribution Moles (based on 1 mole SO₂) S 32.07 g/mol 1 mole O 32.00 g/mol 2 moles - Simplify the ratios: Divide the moles by the greatest common divisor (in this case, 1 for S and 2 for O).
Element Simplest Mole Ratio S 1/1 = 1 O 2/2 = 1 - Write the empirical formula: SO
Thus, the empirical formula for sulfur dioxide is SO.
Practical FAQ
What if my molecular formula has fractions or decimals?
When molecular formulas have fractions, you need to multiply each number by a factor to convert them to whole numbers. For instance, if you have C₃H₆O₃, multiply each subscript by 2 to convert it into C₆H₁₂O₆ (glucose). Then follow the steps outlined to find the empirical formula.
How do I deal with compounds containing polyatomic ions?
For compounds with polyatomic ions, treat the ion as a single entity when calculating moles. For example, in Mg₃(PO₄)₂, treat PO₄ as one unit. Calculate the moles of each element or ion, simplify the ratios, and then find the empirical formula. In this case, the empirical formula would be Mg₃PO₄.
What should I do if I get non-whole numbers when calculating ratios?
If you end up with non-whole numbers after dividing by the GCD, you need to multiply the entire ratio by a number to make all numbers whole. Be careful to multiply all parts of the ratio equally to maintain the proper ratio.
Following these steps and tips, you should now have a clearer understanding of how to decode empirical formulas. Remember, practice makes perfect. The more you apply these techniques to different compounds, the more intuitive the process will become.
With this guide, you’re now equipped with the practical knowledge and steps needed to decode empirical formulas effectively, allowing you to understand the foundational chemistry behind various compounds in an accessible and engaging manner.