Understanding partial pressure is crucial in various scientific and engineering fields, from chemistry and physics to medical and environmental sciences. This concept, fundamental to gas laws and the behavior of gases in mixtures, often perplexes students and professionals alike. However, mastering how to find partial pressure can unlock a deeper comprehension of how gases interact and contribute to overall pressure in a given system.
Key insights box:
Key Insights
- Partial pressure is essential for understanding gas behavior in mixtures.
- Dalton’s Law of Partial Pressures provides the technical basis for calculations.
- Use the ideal gas law to determine individual gas contributions to total pressure.
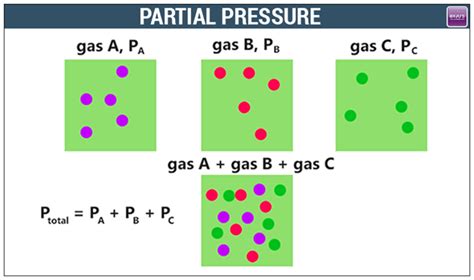
Understanding partial pressure requires a firm grasp of Dalton’s Law of Partial Pressures, which states that the total pressure exerted by a gas mixture is equal to the sum of the partial pressures of each individual gas in the mixture. This insight is practical in contexts like respiratory physiology, where understanding the partial pressure of oxygen and carbon dioxide is crucial for assessing respiratory function.
Dalton’s Law of Partial Pressures
Dalton’s Law is a fundamental principle used to determine the partial pressure of each gas in a mixture. According to the law, the total pressure exerted by a mixture of non-reacting gases is the sum of the partial pressures of each individual gas. Mathematically, this is expressed as P_total = P_1 + P_2 + P_3 +… + P_n, where P_total is the total pressure and P_1, P_2, P_3,…, P_n are the partial pressures of the respective gases. This principle enables precise calculations in various applications, from designing scuba diving equipment to understanding atmospheric conditions.The Ideal Gas Law
The ideal gas law, PV=nRT, integrates the concept of partial pressure in determining individual gas contributions to the total pressure within a mixture. Here, P stands for pressure, V for volume, n for the number of moles, R for the universal gas constant, and T for temperature. To find the partial pressure of a particular gas in a mixture, you isolate its component in the ideal gas law equation. This is particularly useful in laboratory settings where precise gas measurements are essential. For instance, in a controlled experiment involving a mixture of gases, calculating the partial pressure of each gas can reveal insights into their individual behaviors and interactions.FAQ Section
How does humidity affect partial pressure?
Humidity impacts partial pressure by adding water vapor to the gas mixture, which contributes its own partial pressure. This must be accounted for when calculating the partial pressure of other gases in a moist atmosphere.
Can partial pressure be used to predict gas behavior in a closed system?
Yes, partial pressure helps predict gas behavior in closed systems by providing insights into individual gas contributions to the total pressure, which can be vital in understanding gas distribution and interactions under varying conditions.
By delving into the principles of Dalton’s Law of Partial Pressures and utilizing the ideal gas law, one can master the calculation and understanding of partial pressure. This knowledge not only facilitates academic exploration but also enhances practical applications in various scientific and engineering disciplines.