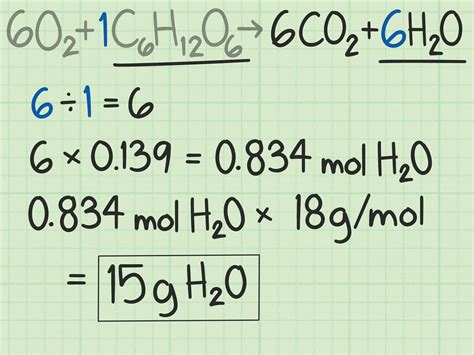Are you struggling to understand the concept of theoretical yield in your chemistry courses or lab work? It can seem like a daunting topic, but fear not—this guide will walk you through every step to make it easy and comprehensible. We’ll cover everything from the basics to some advanced tips, so you can breeze through your assignments and get a solid grasp on this important concept.
Understanding Theoretical Yield: The Basics
The theoretical yield is the maximum amount of product that could be formed from a chemical reaction, based on the initial amount of reactants. Think of it as the ideal scenario where every bit of the starting material converts into the desired product. It’s a key concept in chemistry that helps you predict the outcomes of reactions and understand efficiency.
Why Knowing the Theoretical Yield Matters
Knowing the theoretical yield is essential for several reasons:
- It helps you determine the efficiency of a reaction.
- You can use it to compare the actual yield you obtain in the lab with the ideal yield.
- It forms the foundation for understanding other chemical concepts like percentage yield and limiting reactants.
With this foundational knowledge under your belt, let's dive deeper into calculating the theoretical yield.
Step-by-Step Guide to Calculate Theoretical Yield
To calculate the theoretical yield, follow these steps methodically:
Step 1: Write the Balanced Chemical Equation
Start by writing a balanced chemical equation for the reaction. Balancing ensures that the number of atoms of each element on the reactants side equals the number on the products side.
Step 2: Identify the Limiting Reactant
Next, determine the limiting reactant. This is the reactant that will be completely consumed first and thereby limits the amount of product formed. To do this, compare the mole ratios of the reactants based on their provided quantities.
Step 3: Convert Reactant Quantities to Moles
Use the molar mass of each reactant to convert their given quantities (in grams) to moles. This provides a clear way to compare the amounts and identify the limiting reactant.
Step 4: Calculate the Theoretical Yield
Once you’ve identified the limiting reactant, use stoichiometry to calculate how many moles of product can be formed. Convert the moles of product to the desired units (usually grams).
Quick Reference
Quick Reference
- Immediate action item: Balance the chemical equation for the reaction.
- Essential tip: Convert all given quantities to moles to compare amounts and determine the limiting reactant.
- Common mistake to avoid: Assuming the first reactant listed is always the limiting reactant without verifying.
Detailed How-To Section: Calculating the Theoretical Yield
To illustrate this with a practical example, let’s use a common reaction in organic chemistry—the reaction between ethane (C2H6) and oxygen (O2) to form carbon dioxide (CO2) and water (H2O).
The balanced chemical equation is:
2C2H6 + 7O2 → 4CO2 + 6H2O
Example Step-by-Step Calculation
Let's say you have 5 grams of ethane and 10 grams of oxygen, and you want to find out the theoretical yield of carbon dioxide.
Step 1: Convert Given Quantities to Moles
First, find the molar mass of each reactant:
- Molar mass of C2H6: (2 × 12.01) + (6 × 1.01) = 30.07 g/mol
- Molar mass of O2: (2 × 16.00) = 32.00 g/mol
Next, convert the masses of each reactant to moles:
- Moles of C2H6: 5 g ÷ 30.07 g/mol = 0.166 moles
- Moles of O2: 10 g ÷ 32.00 g/mol = 0.3125 moles
Step 2: Identify the Limiting Reactant
Use the mole ratio from the balanced equation to determine which reactant is limiting. According to the balanced equation, 2 moles of C2H6 react with 7 moles of O2:
- Mole ratio (C2H6 : O2) = 2 : 7
- Calculate the ratio of moles present: 0.166 : 0.3125
- Simplify this ratio by dividing each term by the smaller number, 0.166: (0.166/0.166) : (0.3125/0.166) = 1 : 1.887
Since 1.887 > 7/2 = 3.5, oxygen (O2) is the limiting reactant.
Step 3: Calculate the Theoretical Yield of CO2
Using stoichiometry, determine the amount of CO2 produced from the limiting reactant:
- From the balanced equation, 2 moles of C2H6 produce 4 moles of CO2.
- Thus, 1 mole of C2H6 produces 2 moles of CO2.
- Since we have 0.166 moles of C2H6, the moles of CO2 produced = 0.166 × 2 = 0.332 moles.
Finally, convert moles of CO2 to grams:
- Molar mass of CO2: (12.01 + (2 × 16.00)) = 44.01 g/mol.
- Theoretical yield of CO2: 0.332 moles × 44.01 g/mol = 14.58 g.
Practical FAQ
Why isn’t my actual yield matching the theoretical yield?
When your actual yield doesn’t match the theoretical yield, there are a few common reasons:
- Incomplete reactions: Some reactants might not fully convert into products.
- Loss of product during process: Small amounts of product can be lost during transfer or setup.
- Side reactions: Unintended side reactions might consume some of the reactants.
- Measurement errors: Small errors in measuring reactants or product can lead to discrepancies.
Understanding these potential issues helps you troubleshoot and improve future experiments.