Calculating molar mass is a fundamental concept in chemistry that serves as a cornerstone for various applications in chemical analysis and synthesis. It is essential for determining the quantity of substances in chemical reactions, preparing solutions, and interpreting results from experiments. This guide provides a concise yet comprehensive approach to quickly and easily calculating molar mass, ensuring you can apply this knowledge effectively in your scientific endeavors.
Key Insights
- Understanding molar mass allows for precise measurement in chemical reactions.
- The periodic table is essential for obtaining atomic masses.
- Practice calculating molar mass with real examples.
Understanding Molar Mass
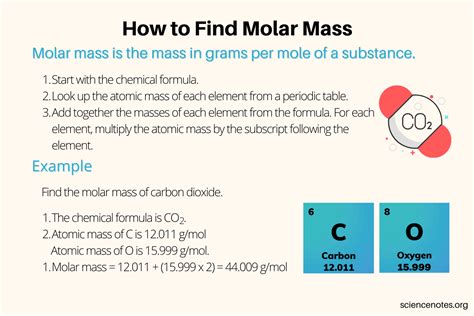
Molar mass, often expressed in grams per mole (g/mol), is the mass of one mole of a given substance. It is a critical component in chemistry because it bridges the gap between the atomic scale and macroscopic quantities. To calculate molar mass, you sum the atomic masses of all atoms in a molecule. Atomic masses are typically found on the periodic table and are provided in atomic mass units (amu). By understanding and using this simple yet powerful concept, you can accurately measure and manipulate substances in your scientific work.Step-by-Step Calculation
Calculating molar mass involves a straightforward process that can be broken into manageable steps. First, identify the chemical formula of the compound. Next, look up the atomic masses of each element in the compound on the periodic table. For example, if you are calculating the molar mass of water (H₂O), you would need to find the atomic masses of hydrogen and oxygen. Hydrogen has an atomic mass of approximately 1.01 g/mol, while oxygen has an atomic mass of approximately 16.00 g/mol. Finally, multiply each atomic mass by the number of its respective atoms in the formula and add these values together. For H₂O, this would be (2 × 1.01 g/mol) + (1 × 16.00 g/mol) = 18.02 g/mol. This process ensures precision in your calculations.Practical Examples
To illustrate the practicality of calculating molar mass, let’s consider a couple of real-world examples. Suppose you need to prepare a solution of sodium chloride (NaCl). To find out how many grams are in one mole of NaCl, you add the atomic mass of sodium (approximately 22.99 g/mol) to the atomic mass of chlorine (approximately 35.45 g/mol), resulting in a molar mass of 58.44 g/mol. This calculation helps you accurately measure the amount of NaCl needed. Another example includes calculating the molar mass of glucose (C₆H₁₂O₆). By summing the atomic masses of six carbon atoms (6 × 12.01 g/mol), twelve hydrogen atoms (12 × 1.01 g/mol), and six oxygen atoms (6 × 16.00 g/mol), you find the molar mass to be 180.16 g/mol. These practical applications underscore the importance of molar mass in everyday chemistry.Can molar mass change for different isotopes of the same element?
No, molar mass calculations are based on the atomic masses listed on the periodic table, which are averages of all naturally occurring isotopes of each element. These averages take into account the relative abundances of each isotope.
What tools can help in molar mass calculations?
Various online calculators and scientific software provide tools to help with molar mass calculations. These can simplify the process, especially for complex molecules, by automatically looking up atomic masses and summing them.
With this guide, you now have a solid understanding of how to calculate molar mass efficiently and accurately. Whether you are working in a laboratory or conducting a theoretical study, mastery of this concept will enhance your chemical calculations and experiments.