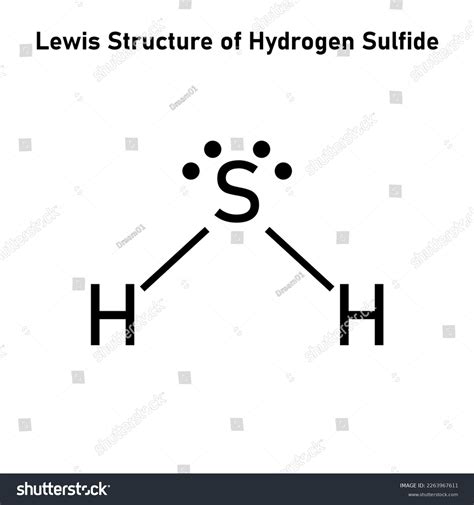
Understanding the Lewis structure of hydrogen sulfide (H₂S) is a critical skill for anyone delving into organic and inorganic chemistry. The molecule, which consists of two hydrogen atoms bonded to a sulfur atom, is more than just a theoretical concept. It holds practical importance in fields such as environmental chemistry and industrial processes. This article breaks down the Lewis structure of H₂S, simplifying it with clear and practical insights.
To start with, the Lewis structure serves as a visual representation of the electrons in a molecule. For H₂S, we need to consider the valence electrons of hydrogen and sulfur. Hydrogen, being in group 1, has one valence electron, while sulfur, in group 16, possesses six valence electrons. Constructing the Lewis structure involves several critical steps that align with the octet rule, ensuring maximum stability.
Key Insights
- Primary insight with practical relevance: H₂S is a molecule that has two lone pairs on sulfur, helping to elucidate its polarity and role in environmental processes.
- Technical consideration with clear application: The tetrahedral geometry of H₂S, derived from its Lewis structure, informs its reactivity and molecular behavior.
- Actionable recommendation: Utilize resonance structures and formal charge calculations to refine your understanding of H₂S’s Lewis structure.
Step-by-Step Breakdown of the Lewis Structure
Let’s delve into the construction of the H₂S Lewis structure step-by-step. First, we identify the least electronegative atom to place in the center—in this case, sulfur. Surrounding it are the two hydrogen atoms. Next, distribute the valence electrons. Sulfur needs eight electrons to achieve a stable octet. Each hydrogen will pair with sulfur using its single valence electron. Initially, sulfur shares one electron pair with each hydrogen atom, forming two single bonds. This accounts for eight electrons, but sulfur needs a total of eight more to achieve a full octet. The remaining two electrons pair up as a lone pair on sulfur, completing its octet.
Understanding Molecular Geometry and Electron Pair Repulsion
Understanding the electron pair repulsion theory is essential in grasping H₂S’s geometry. According to the Valence Shell Electron Pair Repulsion (VSEPR) theory, the two bonding pairs and two lone pairs around the sulfur atom arrange themselves to minimize repulsion, resulting in a tetrahedral shape. Although the molecular shape appears bent due to the lone pairs, this is a simplified view. In reality, the bent geometry can be better understood as derived from the tetrahedral electron pair geometry. This knowledge is crucial when exploring H₂S’s reactivity and interaction in various chemical environments.
What is the formal charge of sulfur in H₂S?
The formal charge on sulfur in H₂S can be calculated by subtracting the number of valence electrons on sulfur in its neutral state (six) from the number of electrons it has in the molecule (four non-bonding electrons plus two it uses for bonding). This results in a formal charge of zero.
Why is H₂S important in environmental chemistry?
Hydrogen sulfide is a key compound in environmental chemistry because it is a byproduct of anaerobic decomposition of organic matter, especially in water-logged environments. It's also a pollutant that contributes to “sour gas” in natural gas. Understanding its Lewis structure helps in predicting its reactivity and devising methods for its removal or neutralization in industrial processes.
This concise guide demystifies the hydrogen sulfide Lewis structure, highlighting its practical applications and chemical significance. From understanding its geometry to recognizing its role in environmental chemistry, mastering this fundamental concept opens the door to more complex chemical studies.