Understanding the polarity of boron trifluoride (BF3) can be a fundamental aspect of comprehending molecular behavior in chemistry. BF3 is a colorless gas with significant applications in various industrial processes, from serving as a catalyst to being involved in chemical synthesis. This article aims to elucidate its polarity through expert insights and practical examples.
The crux of polarity in molecules revolves around electronegativity differences and molecular geometry. Polarity leads to distinct dipole moments, resulting in varying physical properties such as boiling points and solubility. The primary insight is that BF3, despite its triatomic composition, is nonpolar due to its symmetrical trigonal planar structure.
Key Insights
- BF3 is nonpolar despite having polar B–F bonds due to its symmetrical structure.
- Electronegativity differences between boron and fluorine create individual bond dipoles.
- The symmetrical geometry of BF3 results in the cancellation of these dipole moments.
Electronegativity and Bond Polarity
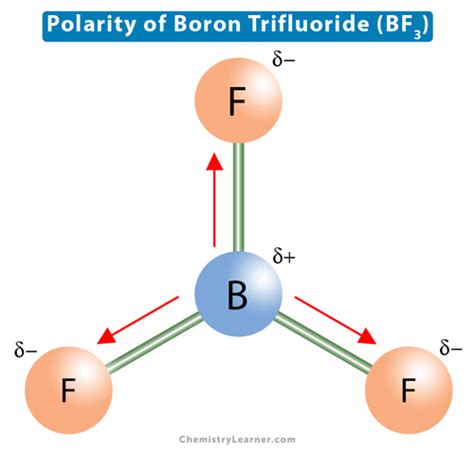
Boron trifluoride comprises three fluorine atoms bonded to a central boron atom. Fluorine’s higher electronegativity compared to boron creates polar B–F bonds. Each B–F bond has a dipole moment pointing away from the boron atom towards the fluorine atom. These bonds are polar because fluorine attracts the shared electrons more strongly than boron, generating a partial negative charge on fluorine and a partial positive charge on boron.Geometrical Symmetry and Molecular Polarity
Although individual B–F bonds are polar, the symmetrical arrangement of the three bonds in a trigonal planar geometry is crucial. In a trigonal planar shape, the bond dipoles point towards the apices of an imaginary triangle surrounding the central boron atom. This geometric configuration leads to the cancellation of individual dipole moments. The symmetrical structure ensures that the vector sum of these dipole moments is zero, resulting in a nonpolar molecule overall.Consider the trigonal planar geometry as if it were a perfect piece of paper with an arrow (dipole) at each corner; the arrows pointing in different directions cause the net dipole to be zero. This symmetrical balance is why BF3 does not exhibit polarity at the molecular level.
Why is BF3 nonpolar if it has polar bonds?
BF3 has polar B–F bonds due to the electronegativity difference between boron and fluorine, but its symmetrical trigonal planar geometry causes the dipole moments to cancel each other out.
What are practical implications of BF3's nonpolarity?
BF3's nonpolarity affects its interaction with other molecules. For example, it does not dissolve in water, a polar solvent, but is more soluble in nonpolar solvents like carbon tetrachloride.
This understanding of BF3’s polarity underscores its practical applications and the behavior of similar molecules in chemical processes. By integrating these scientific insights into practical applications, chemists can better predict and manipulate the behavior of various compounds in both laboratory and industrial settings.