Carbon tetrachloride (CCL4) is a chemical compound that often finds its way into discussions about polarity in organic chemistry. Understanding whether CCL4 is polar or nonpolar can be crucial for a range of applications in both laboratory and industrial settings. The primary goal of this guide is to address the essential aspects of CCL4's polarity, offering a clear, practical, and easy-to-understand explanation for anyone from students to professionals in the field.
Is CCL4 Polar: Unveiling the Quick Insights
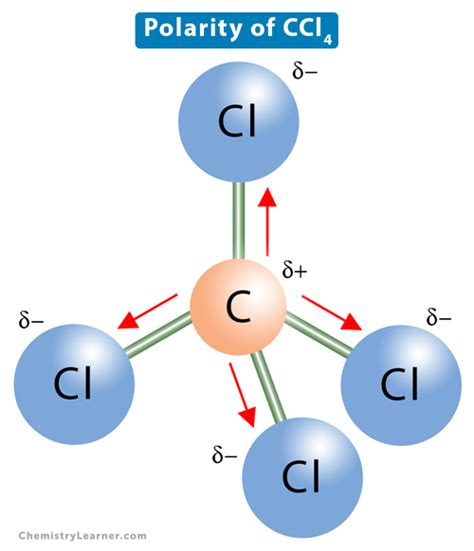
When addressing the question of whether CCL4 is polar, it's important to start with some fundamental principles. Polarity in molecules stems from the difference in electronegativity between the atoms involved in the bonds and the molecular geometry. CCL4, or carbon tetrachloride, consists of one carbon atom bonded to four chlorine atoms. The electrons in each carbon-chlorine bond are not shared equally due to chlorine's higher electronegativity compared to carbon. However, the molecular geometry of CCL4 plays a crucial role.
CCL4 adopts a tetrahedral shape. In a tetrahedral arrangement, the four C-Cl bonds are evenly distributed in space, essentially canceling out any dipole moments, resulting in an overall nonpolar molecule. This symmetry ensures that while individual bonds have dipole moments, they are balanced, leading to a net dipole moment of zero.
Quick Reference
Quick Reference
- Immediate action item with clear benefit: Calculate the electronegativity difference to understand bond polarity.
- Essential tip with step-by-step guidance: Utilize molecular geometry tools to predict molecular polarity.
- Common mistake to avoid with solution: Don’t overlook molecular symmetry when assessing polarity.
Understanding Polarity: A Detailed Exploration
To delve deeper into the polarity of CCL4, we need to consider both the molecular structure and the principles of chemical bonding. Let's break it down step by step:
Electronegativity Differences
The first step in determining polarity is to look at the electronegativity values of the atoms involved. The electronegativity of carbon is 2.55, while that of chlorine is 3.16. The significant difference in these values suggests that there should be a polar bond between carbon and chlorine. However, this alone isn't enough to conclude that the entire molecule is polar.
Molecular Geometry
The molecular geometry of CCL4, which is tetrahedral, is just as important as the electronegativity differences. In a tetrahedral shape, each C-Cl bond vector is positioned to cancel out any dipole moment due to symmetry. This means the bond dipoles create a symmetrical shape that results in an overall nonpolar molecule.
To understand this better, imagine if you place four arrows (representing dipole moments) around a central point (carbon atom) each pointing in such a way that the molecular symmetry is maintained. Even though each arrow points towards a different direction, their equal distribution ensures that they balance each other out, resulting in a net zero dipole moment.
Visual Representation
Using visual aids such as molecular model kits or online molecular geometry tools can be highly beneficial in truly grasping this concept. Seeing how the bonds arrange in three-dimensional space reinforces the idea that even though each individual bond has a dipole moment, the molecule’s overall polarity is zero.
Practical Applications: Why Understanding CCL4 Polarity Matters
Understanding the polarity of CCL4 is not just an academic exercise. It has practical implications in various fields including chemistry, pharmaceuticals, and environmental science.
For instance, knowing that CCL4 is nonpolar helps in predicting its solubility characteristics, as nonpolar substances typically dissolve well in nonpolar solvents but poorly in polar ones. This knowledge is crucial when designing extraction procedures or when formulating solvents for chemical reactions.
Practical Example: Using CCL4 in Solvent Systems
Let’s consider a real-world example where understanding CCL4’s polarity could be valuable: in pharmaceutical extraction processes. If a drug compound needs to be extracted from an organic matrix, knowing that CCL4, being nonpolar, will dissolve nonpolar substances while leaving polar components behind can guide the choice of solvent.
For example, if you need to extract nonpolar alkaloids from plant material, CCL4 would be an excellent choice because it won’t dissolve polar sugars or other polar components of the plant, thus purifying the desired compound.
FAQs on CCL4 Polarity
Why is CCL4 Considered Nonpolar?
CCL4 is considered nonpolar primarily because of its tetrahedral molecular geometry. Even though each carbon-chlorine bond has a dipole moment due to the electronegativity difference between carbon and chlorine, the symmetry of the tetrahedral shape ensures that these dipoles cancel each other out. This results in an overall nonpolar molecule.
Can Polar Solvents Dissolve CCL4?
CCL4, being nonpolar, does not dissolve well in polar solvents. Polar solvents, such as water or alcohol, have a higher affinity for polar molecules due to their own dipole moments. Since CCL4 lacks significant polarity, it is more likely to dissolve in nonpolar solvents like hexane or benzene. This characteristic can be used to separate CCL4 from polar substances during purification processes.
How Does Symmetry Affect Polarity?
Symmetry plays a crucial role in determining molecular polarity. Even if individual bonds in a molecule are polar, if the molecule's shape is symmetrical in such a way that the dipoles cancel each other out, the molecule will be nonpolar. For instance, in CCL4, the tetrahedral arrangement means that each C-Cl bond vector is equally distributed in space, leading to a net zero dipole moment. This concept is essential in understanding why some symmetrical tetrahedral molecules like CCL4 are nonpolar despite having polar bonds.
By understanding the nonpolar nature of CCL4, professionals can make informed decisions about its use in various applications ranging from chemical synthesis to extraction processes in pharmaceuticals. This knowledge not only aids in theoretical understanding but also provides practical solutions in applied chemistry settings.