Unveiling the Polarity of Hydrogen Cyanide: A Deep Dive
To understand the polarity of hydrogen cyanide (HCN), we need to explore its molecular structure and the distribution of electron density. Hydrogen cyanide is a small, linear molecule comprised of hydrogen, carbon, and nitrogen atoms. This molecule’s polarity plays a significant role in its chemical behavior, reactivity, and interactions with other substances. The insights derived from understanding the polarity of HCN have practical implications in various fields, including chemistry, environmental science, and industrial applications.
Key Insights
- Hydrogen cyanide (HCN) exhibits a polar nature due to its linear molecular structure and the electronegativity difference between nitrogen and carbon atoms.
- The dipole moment arises from the significant electronegativity difference between nitrogen (3.04) and carbon (2.55), creating a polarized bond.
- Understanding HCN's polarity is essential for predicting its reactivity and solubility in various solvents.
Molecular Structure and Electronegativity
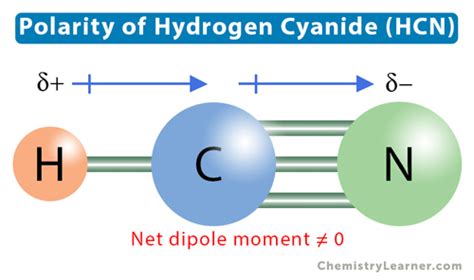
The molecular structure of HCN is linear, with the hydrogen atom bonded to the carbon atom, which in turn is bonded to the nitrogen atom. This arrangement leads to an uneven distribution of electrons due to the substantial difference in electronegativity between nitrogen and carbon. Nitrogen, being more electronegative, pulls electrons more tightly, resulting in a partial negative charge (δ-) on the nitrogen atom and a partial positive charge (δ+) on the carbon atom.
Dipole Moment and Polarity
The dipole moment of a molecule quantifies its polarity and is a vector that points from the partial positive charges to the partial negative charges. In HCN, the significant electronegativity difference between nitrogen (3.04) and carbon (2.55) creates a strong dipole moment. This polarity influences many of HCN’s physical properties. For instance, HCN is more soluble in polar solvents such as water and other polar organic solvents due to the favorable interaction between the polar HCN molecule and the polar solvent molecules.
In practical terms, the polarity of HCN affects its chemical reactivity, particularly in nucleophilic substitution reactions where it can act as both a nucleophile and an electrophile. The polarized bond in HCN makes it a versatile reagent in various organic synthesis processes.
How does the polarity of HCN affect its interactions with other molecules?
The polarity of HCN enhances its ability to interact with other polar molecules through dipole-dipole interactions and hydrogen bonding, which is crucial in determining its solubility in polar solvents and its role in chemical reactions.
What are the practical applications of understanding HCN's polarity?
Understanding HCN's polarity is vital for predicting its behavior in different environments, enhancing its use in organic synthesis, and ensuring its safe handling and storage in industrial and research settings.
The polarity of hydrogen cyanide reveals much about its fundamental properties and potential applications. The linear molecular structure and electronegativity differences give rise to a pronounced dipole moment, making HCN a polar molecule with unique interactions and reactivity patterns. This knowledge serves as a foundation for both theoretical studies and practical applications in various scientific and industrial domains.