Have you ever wondered what lies beneath the surface of Lewis diagrams, especially when it comes to complex molecules like nitrogen dioxide (N2O)? In this practical guide, we’ll decode the Lewis diagram of N2O, addressing common questions, offering actionable advice, and providing clear examples to ensure you get the most out of this knowledge. Whether you’re a student trying to grasp this concept or a professional looking to expand your understanding, this guide is tailored to meet your needs and help you master the Lewis diagram of N2O with ease.
Understanding the Lewis Diagram of N2O
The Lewis diagram for N2O, or nitrous oxide, might seem intimidating at first, but with some straightforward steps and a bit of practice, you’ll find it becomes an invaluable tool for understanding molecular bonding and structure.
Here's why mastering the Lewis diagram for N2O is important:
- It helps you visualize the electron distribution around nitrogen and oxygen atoms.
- Understanding the diagram aids in predicting the molecule's chemical properties and reactivity.
- It forms the basis for comprehending more complex molecules and their interactions.
Quick Reference
Quick Reference
- Immediate action item: Identify valence electrons for each atom in N2O.
- Essential tip: Place the nitrogen atoms on either side of the oxygen atom and connect them with a bond.
- Common mistake to avoid: Failing to distribute lone pairs correctly can lead to an incorrect Lewis structure.
Step-by-Step Guide to Drawing the Lewis Diagram for N2O
To draw the Lewis diagram for N2O, let’s break it down step by step. Here’s how you can approach it:
Step 1: Determine the Total Number of Valence Electrons
Start by identifying the valence electrons of each atom involved:
| Atom | Valence Electrons |
|---|---|
| Nitrogen (N) | 5 each for two nitrogen atoms |
| Oxygen (O) | 6 |
So, the total number of valence electrons for N2O is:
2(5) + 6 = 16 valence electrons
Step 2: Place the Atoms and Connect Them
In N2O, we have two nitrogen atoms on either side of the oxygen atom:
N—O—N
Connect them with a single bond:
N—O—N
Each nitrogen atom shares its electron with the oxygen atom, forming a covalent bond:
Step 3: Distribute Remaining Electrons
Next, we need to distribute the remaining valence electrons around the atoms:
- Calculate the remaining electrons: 16 - 2 (used for the single bond) = 14 electrons
- Distribute the remaining electrons in pairs around each atom to satisfy the octet rule (except for hydrogen which follows the duet rule). Start with the oxygen atom because it is more electronegative and will hold lone pairs.
Here’s a breakdown:
| Atom | Electrons Assigned |
|---|---|
| Each nitrogen | 3 lone pairs (6 electrons) |
| Oxygen | 2 lone pairs (4 electrons) |
Step 4: Check the Octet Rule
Ensure each atom has a complete octet (or duet for hydrogen). After placing the lone pairs:
| Atom | Electron Configuration |
|---|---|
| Nitrogen (N) | 8 electrons (2 bonds + 3 lone pairs) |
| Oxygen (O) | 8 electrons (2 bonds + 2 lone pairs) |
All atoms now have a complete octet, and the Lewis diagram for N2O is complete.
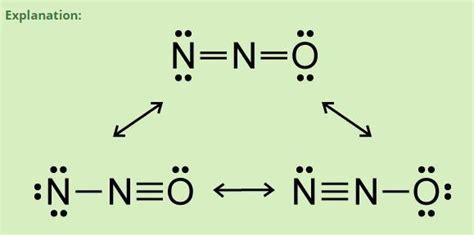
Step 5: Resonance Structures
While N2O has a primary structure, it can also exhibit resonance. Consider these alternative representations:
- Switch the lone pairs between the nitrogen atoms and the oxygen to form multiple bonds:
- N=O—N
- N—O=N
These structures help us understand different electronic configurations, even though one is predominant.
Practical Applications of N2O Lewis Diagram
Understanding the Lewis diagram for N2O is more than academic; it has practical applications:
- It helps in predicting the chemical behavior of nitrous oxide in various reactions.
- It provides insight into its role as an anesthetic and a greenhouse gas.
- It assists in environmental science by understanding its contribution to climate change.
FAQ Section
Why is the Lewis diagram important for N2O?
The Lewis diagram for N2O is important because it provides a clear picture of how electrons are shared between the nitrogen and oxygen atoms. This understanding is crucial for predicting the molecule’s reactivity, bonding patterns, and overall behavior in various chemical reactions. Moreover, it helps in grasping the molecule’s role in different applications, such as medical uses and environmental impact.
How does N2O differ from other nitrogen oxides?
N2O, or nitrous oxide, differs from other nitrogen oxides in its molecular structure and reactivity. While compounds like NO (nitric oxide) and NO2 (nitrogen dioxide) have different bonding characteristics and uses, N2O’s Lewis diagram shows a unique arrangement with nitrogen atoms bonded to oxygen. This structure results in different physical and chemical properties. For example, N2O is less reactive than NO2 and has different applications, such as its use as an anesthetic in medical procedures.
What are common mistakes when drawing the Lewis diagram for N2O?
Common mistakes when drawing the Lewis diagram for N2O include miscalculating the number of valence electrons, incorrectly placing lone pairs, and misunderstanding the resonance structures. It’s essential to correctly identify the valence electrons, place the atoms in the appropriate structure, and ensure the octet rule is satisfied. Missteps often occur in the placement of lone pairs, leading to an incorrect representation of the molecule’s electron distribution.
Conclusion
Mastering the Lewis diagram of N2O is a vital step toward understanding molecular chemistry. With this guide, you now have a step-by-step approach to drawing the Lewis structure, identifying electron distribution, and understanding its applications. By addressing common questions and tackling potential challenges, this guide aims to provide you with a robust understanding that extends beyond mere academic requirements.
Armed with this knowledge, you can apply these principles to a variety of contexts, from classroom exercises to real-world chemical processes. Keep practicing and exploring, and soon the intricacies of the N2O Lewis diagram will become second nature!