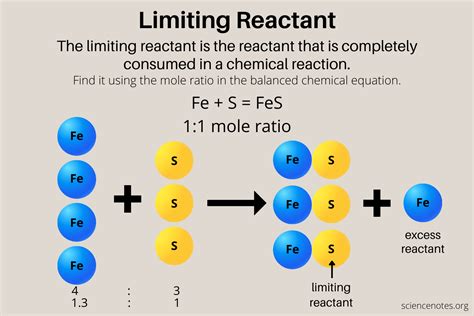
Understanding the concept of limiting reactants is pivotal for anyone involved in chemical reactions, whether you are a seasoned chemist or a student delving into organic chemistry for the first time. The limiting reactant is the substance that determines the amount of product formed during a chemical reaction, as it is entirely consumed first. By identifying and understanding the limiting reactant, chemists can accurately predict yields and manage resources effectively.
Key Insights
Key Insights
- Primary insight with practical relevance: Recognizing the limiting reactant is essential for maximizing product yield in both laboratory and industrial settings.
- Technical consideration with clear application: Calculating stoichiometric ratios helps determine the limiting reactant, ensuring precise control over reaction conditions.
- Actionable recommendation: Always perform a limiting reactant analysis to optimize reaction efficiency and minimize waste.
Fundamental Concept of Limiting Reactants
To comprehend the role of limiting reactants, we must first delve into stoichiometry. In a chemical reaction, reactants combine in defined ratios. The reactant that is completely used up first, and thus limits the amount of product formed, is identified as the limiting reactant. For instance, in the reaction of hydrogen with oxygen to form water, if there is more hydrogen than oxygen, hydrogen will be the limiting reactant, as it will be fully consumed before oxygen does.Calculation of Limiting Reactants
Determining the limiting reactant involves comparing the amount of reactants present using their stoichiometric coefficients from the balanced chemical equation. Suppose we have a reaction where 2 moles of A react with 3 moles of B to form a product. If we have 4 moles of A and 6 moles of B, we compare their ratios:- The ratio of A to its required amount in the balanced equation: 4 moles A / 2 = 2
- The ratio of B to its required amount: 6 moles B / 3 = 2
From this calculation, we can see that both reactants are present in stoichiometric proportions. To determine which is limiting, consider the actual amounts: if only 4 moles of A are available but 6 moles of B are needed based on the provided amounts, then A is the limiting reactant.
Practical Application in Industrial Chemistry
In industrial chemistry, recognizing the limiting reactant is not just an academic exercise but a critical operational factor. Companies producing bulk chemicals must meticulously calculate the limiting reactants to ensure cost-efficiency and maximum yield. For example, in the manufacture of ammonia using the Haber process, nitrogen and hydrogen are combined in specific ratios. Misidentifying the limiting reactant can lead to underproduction or the wastage of expensive reactants, significantly impacting profitability.FAQ Section
How does knowing the limiting reactant help in real-world applications?
Knowing the limiting reactant helps in optimizing resource use and maximizing yield in both laboratory and industrial processes. It ensures efficient production and minimizes waste, leading to cost savings and better resource management.
Can a reaction have more than one limiting reactant?
Typically, in a straightforward chemical reaction, there is one limiting reactant. However, in more complex reactions with multiple steps or in systems where reactants are added in stages, multiple reactants might appear as limiting at different stages, necessitating a re-evaluation of the limiting reactant as the reaction progresses.
In conclusion, mastering the identification of limiting reactants is essential for anyone aiming to excel in chemistry. This knowledge enhances precision in laboratory settings and is invaluable in industrial applications where efficiency and cost-effectiveness are paramount. The next time you engage in a chemical reaction, take a moment to identify your limiting reactant—it could make all the difference in your outcome.