Converting liters to grams can be an essential skill for both scientific and culinary applications. While both liters and grams are units of measurement, they apply to different types of measurement: liters for volume and grams for mass. The conversion requires knowing the density of the substance in question since density determines mass per unit volume. This guide aims to clarify the approach to convert liters to grams effectively.
The first step in converting liters to grams is to understand the relationship between volume and mass. Density is the key factor here; it measures mass per unit volume (usually in g/cm³ or kg/L). With the density in hand, you can determine how much mass corresponds to a given volume. Let’s dive into practical insights and specific examples to elucidate this conversion process.
Key Insights
- Primary insight with practical relevance: Understanding and using the density of a substance is critical for converting liters to grams.
- Technical consideration with clear application: The formula mass (g) = volume (L) × density (g/mL) is straightforward but hinges on knowing the density.
- Actionable recommendation: Always determine the density of your substance before attempting to convert liters to grams.
Understanding Density
Density is a fundamental property in chemistry and physics. It describes how much mass is contained within a certain volume. The formula for density is density = mass/volume. In practice, you often need to convert from volume to mass using the density. For example, if you have water, which has a density of 1 gram per milliliter (g/mL) or 1000 grams per liter (g/L), converting is straightforward: simply multiply the volume in liters by 1000.Practical Conversion Example
To convert a volume measured in liters to grams, follow this step-by-step procedure:- Determine the density of the substance you're dealing with. For water, it’s 1 g/mL. For other substances like honey or oil, this number will differ.
- Convert liters to milliliters if necessary, knowing that 1 liter = 1000 milliliters.
- Apply the formula: mass (g) = volume (L) × density (g/mL). For water, this would be mass (g) = volume (L) × 1000 g/L.
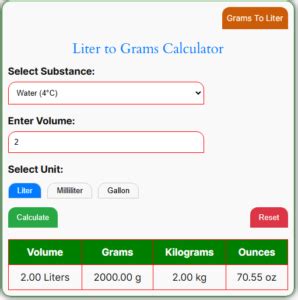
- Calculate the mass. For example, to convert 2 liters of water to grams, you multiply 2 liters by 1000 grams per liter, resulting in 2000 grams.
Real-World Application
In a culinary context, this conversion is essential when following precise recipes. Suppose you are baking a cake that requires 0.5 liters of honey. Knowing honey’s density is approximately 1.42 g/mL, you can convert this to grams by calculating mass (g) = 0.5 L × 1420 g/L, resulting in 710 grams of honey needed. In scientific settings, accurate conversions are crucial for chemical reactions, ensuring precise measurements that impact the outcomes of experiments.What if the density is not readily available?
If you don’t have the density readily available, consult a reliable source or database. Online resources and scientific literature often provide density values for various substances.
Can this method be used for gases?
Yes, but gas densities vary significantly with temperature and pressure. Use the ideal gas law or specific gas density tables for accurate conversions.
In conclusion, converting liters to grams is a straightforward yet nuanced process that hinges on understanding and utilizing the density of the substance. Whether you’re a scientist, a chef, or simply someone needing to make precise conversions, having a firm grasp on these principles ensures accuracy and success in your applications.