Understanding the lithium proton number is essential for comprehending the foundational aspects of atomic physics. This concept is particularly relevant for scientists, educators, and students delving into the realm of atomic structure and nuclear chemistry. At its core, the lithium proton number reflects the identity of the element and plays a pivotal role in various chemical reactions and applications. Let’s embark on a journey to decode the intricacies of this atomic detail.
Key Insights
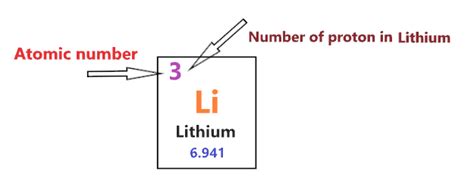
- The lithium proton number is 3, signifying the number of protons in the nucleus of a lithium atom.
- This characteristic is crucial for determining the element’s identity and behavior in chemical reactions.
- Understanding the lithium proton number helps in the exploration of its practical uses in technology and medicine.
Atomic Structure of Lithium
Lithium, denoted by the symbol Li, occupies position 3 on the periodic table, thus having a lithium proton number of 3. This number indicates that every lithium atom contains three protons in its nucleus. The atomic number, which correlates directly with the number of protons, establishes lithium as a distinct element. The nucleus also contains neutrons; however, the proton count remains a primary identifier.In the atomic nucleus, protons carry a positive charge, whereas electrons, which orbit the nucleus, carry a negative charge. A neutral lithium atom has an equal number of protons and electrons—three of each. This balance is fundamental in determining that lithium is neither positively nor negatively charged in its natural state.
Chemical Behavior and Reactions
The lithium proton number significantly influences the chemical behavior of lithium. With only three protons, lithium’s small nucleus and electron configuration lead to unique chemical properties. It’s notably reactive, especially with water, forming lithium hydroxide and hydrogen gas. This reactivity is due to the relatively low ionization energy required to remove an electron from its outer shell.Lithium’s chemical properties extend into its uses in batteries, where it forms the active component in lithium-ion batteries. These batteries leverage the lithium ion’s small size and high mobility, ensuring efficient energy storage and transfer. The consistent and predictable reactions of lithium make it indispensable in advanced technologies, from portable electronics to electric vehicles.
What is the significance of the lithium proton number in its nuclear applications?
The lithium proton number of 3 is crucial in nuclear applications, particularly in nuclear reactors where isotopes like lithium-6 are used as neutron moderators. This function is pivotal in controlling the rate of nuclear reactions, ensuring stability and safety in power generation.
How does the lithium proton number affect its role in medicinal chemistry?
In medicinal chemistry, lithium's proton number dictates its behavior in biological systems. Its ions are utilized in pharmaceuticals, especially in treatments for bipolar disorder. Understanding its atomic identity helps in designing more effective therapeutic compounds.
In conclusion, the lithium proton number, standing at 3, is fundamental to the element’s identity and its wide array of applications. From driving technological advancements to contributing to medical breakthroughs, lithium’s unique atomic characteristics cannot be overstated.