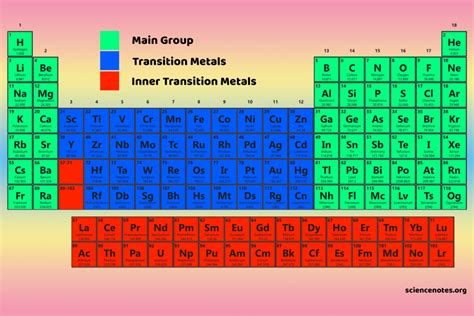
Understanding the essential main group elements in the periodic table is crucial for comprehending fundamental chemistry. These elements form the backbone of chemical reactions and biological processes, thus playing a vital role in various fields from medicine to industrial chemistry. The main group elements are those found in groups 1, 2, and 13 through 18. These elements exhibit unique properties and reactivity patterns that make them indispensable in scientific exploration and application.
Key Insights
- Primary insight with practical relevance: Main group elements are central to organic chemistry and biochemistry.
- Technical consideration with clear application: Understanding electronegativity and ionization energy of main group elements is critical for predicting reaction outcomes.
- Actionable recommendation: Focus on mastering the properties and uses of elements like carbon, oxygen, and nitrogen to enhance your understanding of chemical behaviors.
The Significance of Main Group Elements
Main group elements are pivotal due to their high electronegativity and diverse reactivity. They form the basis for a wide array of compounds that underpin both inorganic and organic chemistry. For instance, carbon (group 14) is fundamental to organic chemistry, enabling the creation of complex molecules essential to life. Similarly, oxygen (group 16) is critical for biological processes such as respiration and energy production in living organisms. Mastery of the chemistry of main group elements allows scientists to innovate and engineer new materials, medicines, and technologies.Reactivity Patterns of Main Group Elements
Each main group element exhibits specific reactivity patterns that are influenced by their electron configuration. Elements in group 1, known as alkali metals, are highly reactive due to their single valence electron which they readily lose to achieve a stable electron configuration. For example, sodium reacts vigorously with water to form sodium hydroxide and hydrogen gas. On the other hand, elements in group 17, the halogens, are highly electronegative and tend to gain one electron to complete their valence shell. Chlorine’s reactivity with metals is a testament to its strong tendency to form salts. Understanding these reactivity patterns is fundamental for predicting and controlling chemical reactions.Why are main group elements so important in biological systems?
Main group elements like carbon, hydrogen, nitrogen, and oxygen are essential for the structure and function of biomolecules. These elements form the backbone of proteins, nucleic acids, carbohydrates, and lipids, which are fundamental to life processes.
How does electronegativity impact the chemistry of main group elements?
Electronegativity influences how main group elements form bonds with other atoms. Higher electronegativity, as seen in elements like fluorine, means that these atoms can attract electrons more strongly, leading to polar covalent bonds or ionic interactions.
The journey through the periodic table reveals the intricate and often surprising behaviors of the main group elements. As we delve deeper into their unique properties and applications, we unlock a world of possibilities that span from the smallest cellular processes to the largest technological advancements.