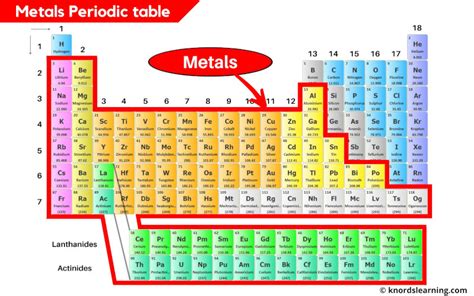
Understanding the properties and behaviors of metals on the periodic table is essential for various scientific, industrial, and technological advancements. Metals play a crucial role in shaping our modern world, from the engines in our cars to the advanced electronics that power our daily lives. This article delves into the fundamental characteristics of metals, provides practical insights, and integrates evidence-based statements with real-world examples to demystify their complexity.
Key Insights
- Primary insight with practical relevance: Metals are vital in construction and manufacturing due to their unique properties such as strength, conductivity, and malleability.
- Technical consideration with clear application: The choice of metal can greatly affect the efficiency and longevity of materials used in aerospace engineering.
- Actionable recommendation: Investing in education and research into advanced metallurgy can lead to innovations in creating stronger, more durable, and more sustainable metals.
The periodic table offers a comprehensive guide to understanding various elements, with metals comprising a significant portion. These elements are typically located on the left side of the periodic table and are characterized by their high density, ductility, malleability, thermal, and electrical conductivity. For instance, iron is a widely used metal due to its ability to be alloyed with carbon to form steel, which is integral in construction. Metals such as aluminum and titanium are prized for their strength-to-weight ratios, making them indispensable in aerospace engineering.
Properties of Metals on the Periodic Table
Metals possess a distinct set of physical and chemical properties that define their usefulness in various applications. Their high melting and boiling points, luster, and ability to form cations are fundamental. For example, copper, with its excellent electrical conductivity, is essential in the production of electrical wiring. Gold’s resistance to corrosion and tarnish makes it ideal for jewelry and high-tech electronics where stability is paramount. Understanding these properties enables scientists and engineers to select the right metals for specific applications, ensuring optimal performance and durability.
Industrial Applications of Metals
The industrial applications of metals are vast and varied, spanning from automotive manufacturing to electronics. In the automotive industry, metals like steel and aluminum are prevalent due to their strength and lightweight properties. Advanced manufacturing techniques like hydroforming and additive manufacturing have further optimized the use of metals in producing safer and more fuel-efficient vehicles. In the electronics sector, metals such as silver and gold are utilized for their exceptional electrical conductivity, allowing for the creation of high-performance circuits and components. Additionally, ongoing research into rare and less commonly used metals, such as palladium and scandium, continues to uncover new possibilities in various high-tech fields.
What makes metals such a crucial part of modern technology?
Metals are indispensable in modern technology due to their unique properties like high strength, electrical conductivity, and thermal conductivity. They are fundamental in constructing electronic devices, machinery, and infrastructure.
How do advancements in metallurgy impact everyday life?
Advancements in metallurgy lead to the development of new alloys with improved characteristics such as increased strength, resistance to corrosion, and better thermal management, enhancing the efficiency and longevity of everyday products.
The study and application of metals on the periodic table are indispensable for progressing various fields. The intrinsic properties of metals drive innovation across industries, making informed knowledge about their characteristics crucial for scientific and engineering advancements.