Monoprotic acids play an integral role in modern chemistry and hold a crucial place in a variety of scientific and industrial applications. Understanding their properties and applications can help chemists, students, and professionals alike optimize their use in various processes. This guide will provide you with a clear, step-by-step approach to comprehending the role of monoprotic acids, addressing common problems and solutions while delivering practical advice to tackle real-world challenges.
Understanding Monoprotic Acids: An Introduction
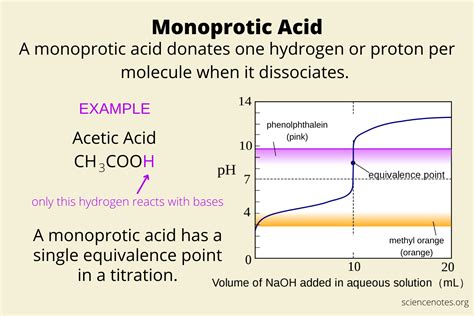
Monoprotic acids are a class of acids that can donate one proton (H+) per molecule during the process of dissociation in a solvent such as water. Examples include hydrochloric acid (HCl), nitric acid (HNO3), and sulfuric acid (H2SO4, when considered in its first dissociation step). They are fundamental in both theoretical chemistry and practical applications like titrations, reactions, and numerous industrial processes. Their role is vital in various domains from pharmaceuticals to environmental chemistry.
Quick Reference
Quick Reference
- Immediate action item with clear benefit: Start by identifying the monoprotic acid involved in your experiment and ensure its concentration and purity are within required specifications.
- Essential tip with step-by-step guidance: To determine the strength of a monoprotic acid, conduct a titration using a standard base (like NaOH) and record the volume at the equivalence point to calculate the acid’s concentration.
- Common mistake to avoid with solution: Avoid underestimating the potential hazards of handling strong monoprotic acids; always use appropriate personal protective equipment (PPE) and work under a fume hood to prevent exposure.
Deep Dive into Handling Monoprotic Acids
Working with monoprotic acids requires both understanding their chemical behavior and adhering to safety protocols. Below we break down the detailed steps for handling and using monoprotic acids effectively.
Safety First: Handling Monoprotic Acids
When dealing with monoprotic acids, especially strong ones like hydrochloric or sulfuric acid, safety should always be your top priority. Here are the crucial steps:
-
Always wear appropriate personal protective equipment (PPE). This includes lab coats, safety goggles, and gloves made of materials resistant to acid exposure.
-
Conduct experiments in a well-ventilated area, preferably under a fume hood, to avoid inhalation of harmful fumes.
-
Use acid-resistant equipment and ensure spill containment measures are in place. For any unexpected spill, neutralize the acid with a suitable base (like sodium bicarbonate) and clean up promptly.
Titration Procedures: Using Monoprotic Acids
Titration is a common technique to determine the concentration of a monoprotic acid. Here’s how to perform it accurately:
-
Begin with a clean burette. Fill it with the monoprotic acid of interest, ensuring there are no air bubbles at the top.
-
Use a pipette to deliver a known volume of the acid solution into an Erlenmeyer flask. Record the initial volume in the burette accurately.
-
Add a few drops of an acid-base indicator (such as phenolphthalein) that will change color at the equivalence point.
-
Slowly titrate with a standard base solution (like sodium hydroxide) while continuously stirring the acid solution. Observe the color change of the indicator.
-
Once the endpoint is reached (indicated by a permanent color change), stop adding the base and record the final volume in the burette.
-
Calculate the concentration of the acid using the formula:
Concentration = (Volume of base used x Concentration of base) / Volume of acid
Applications of Monoprotic Acids in Industry
Monoprotic acids are extensively used in various industrial processes. Here are some key applications:
-
In pharmaceuticals: Monoprotic acids are used in the synthesis of various drugs, where precise control of pH is required.
-
In agriculture: Acids like sulfuric are used to adjust the pH of soils to promote better crop growth.
-
In manufacturing: They are used in the production of metals, dyes, and polymers where pH control is essential.
-
In cleaning agents: Strong monoprotic acids like hydrochloric acid are used in descaling and cleaning industrial equipment.
Practical FAQ
What precautions should be taken while handling monoprotic acids?
When handling monoprotic acids, it’s crucial to take several precautions to ensure your safety and the accuracy of your experiments. Here are specific steps:
-
Always wear appropriate PPE including gloves, goggles, and lab coats. Ensure that the gloves are acid-resistant.
-
Perform experiments under a fume hood to avoid inhalation of harmful vapors.
-
Be cautious of direct contact with skin and eyes. If contact occurs, rinse immediately with plenty of water.
-
Store acids in labeled, secure containers away from incompatible substances.
-
Have neutralizing agents on hand in case of spills.
By following these steps, you can handle monoprotic acids safely and efficiently, minimizing risks and maintaining the integrity of your experiments.
How do you neutralize a monoprotic acid spill?
To neutralize a monoprotic acid spill, it’s essential to act quickly and responsibly. Here are the steps you should follow:
-
Evacuate the area and ensure no one is in harm’s way.
-
Wear PPE before approaching the spill.
-
Use an acid-neutralizing agent such as sodium bicarbonate. Do not use calcium carbonate as it can produce carbon dioxide gas, which may complicate the situation.
-
Apply the neutralizing agent over the spill slowly, allowing it to react with the acid. Use a brush if necessary to spread it evenly.
-
Once the acid has been neutralized, clean the area thoroughly with water. Ensure there is no residual acid left.
Following these steps ensures the safe and effective neutralization of a monoprotic acid spill.
Understanding and working with monoprotic acids effectively involves a mix of theoretical knowledge and practical skills. From titration techniques to safety protocols and industry applications, this guide covers essential areas to ensure your work with monoprotic acids is both safe and effective. Follow the steps, adhere to safety guidelines, and you’ll be well-equipped to handle these crucial chemicals with confidence.