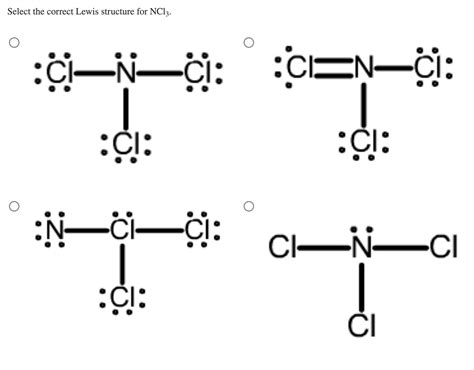
Understanding the Lewis structure of an element or compound is critical for grasping its chemical properties, bonding, and reactivity. This guide dives into the secrets of NCl3's Lewis structure, offering you step-by-step guidance, practical examples, and actionable advice to deepen your comprehension. Whether you are a student or a professional in the field, this guide will empower you with the knowledge needed to master the NCl3 Lewis structure.
Understanding the Need for a Clear Lewis Structure
When we look at the NCl3 molecule, we aim to create a visual representation that reveals the arrangement of atoms and electrons. A clear Lewis structure allows us to predict molecular shape, reactivity, and the types of bonds that exist. Without this understanding, predicting how NCl3 will interact with other substances can be challenging. Let’s break down why understanding the Lewis structure is fundamental for grasping NCl3.
Imagine you are an environmental scientist trying to understand how NCl3 behaves in an industrial setting. Without a thorough understanding of its Lewis structure, predicting its reaction with water or other chemicals could lead to unsafe conditions. A comprehensive grasp helps ensure safety and efficiency in both laboratory and industrial environments.
Quick Reference
Quick Reference
- Immediate action item: Count the total valence electrons for NCl3.
- Essential tip: Use the octet rule to guide your electron placement.
- Common mistake to avoid: Forgetting to minimize formal charges.
Step-by-Step Guide to Drawing NCl3 Lewis Structure
Here’s a step-by-step guide to constructing the NCl3 Lewis structure. This process ensures that you accurately depict the molecule’s electron configuration, which is crucial for understanding its properties and behavior.
Step 1: Count Valence Electrons
The first step in drawing the Lewis structure for NCl3 is determining the total number of valence electrons. Nitrogen has 5 valence electrons, and each chlorine atom contributes 7. Since there are three chlorine atoms, we calculate as follows:
| Element | Valence Electrons | Total |
|---|---|---|
| N | 5 | |
| Cl | 7 x 3 = 21 | |
| Total | 5 + 21 = 26 |
So, the total number of valence electrons available is 26.
Step 2: Place the Central Atom
Nitrogen, being the least electronegative element, will be the central atom. We place nitrogen in the center and surround it with three chlorine atoms.
Step 3: Form Single Bonds
We connect nitrogen to each chlorine atom with a single bond. Each bond uses 2 electrons, so for three bonds, we use 6 electrons. This accounts for 6 out of our 26 valence electrons.
Step 4: Distribute Remaining Electrons
With 20 electrons remaining (26 total – 6 used in bonds), we place these electrons around the chlorine atoms to satisfy their octets. Each chlorine atom initially needs 8 electrons:
1. Start with the first chlorine atom, placing three lone pairs (6 electrons) around it. 2. Repeat this for the second and third chlorine atoms.
At this stage, each chlorine atom has an octet, but nitrogen only has 8 electrons, not fulfilling the octet rule for nitrogen directly. To resolve this, we'll need to redistribute electrons:
Step 5: Minimize Formal Charges
To ensure the most stable Lewis structure, we minimize formal charges. To do this, convert lone pairs on chlorine atoms into bonds with nitrogen, if it makes formal charges more negative or zero. This process might require trial and error to find the most stable configuration:
Here’s how to convert lone pairs into bonds:
- First, look at nitrogen. It needs two more electrons to complete its octet.
- Convert one lone pair from each of the chlorine atoms into a bond with nitrogen.
This approach ensures all atoms achieve an octet:
- Nitrogen will share two electrons from each chlorine, completing its octet.
- Each chlorine will now have three bonds (one with nitrogen and two lone pairs), fulfilling the octet rule.
This results in the final Lewis structure of NCl3, where nitrogen has a formal charge of 0, and each chlorine has a formal charge of -1.
Practical Examples and Application
Let’s delve into real-world examples that illustrate the application of the NCl3 Lewis structure. Understanding these examples can deepen your comprehension and highlight the importance of this concept.
Example 1: Predicting Molecular Geometry
Knowing the Lewis structure of NCl3 helps predict its molecular geometry. The nitrogen atom is surrounded by three chlorine atoms and has one lone pair of electrons. This arrangement corresponds to a trigonal pyramidal geometry, which can be deduced from the electron groups around nitrogen:
- Three bonding pairs
- One lone pair
Example 2: Reaction with Water
To understand how NCl3 reacts with water, knowing its Lewis structure is crucial. When NCl3 encounters water, it undergoes hydrolysis:
NCl3 + 3H2O → H3N-Cl + 3HCl
This reaction helps you understand the behavior of NCl3 in aqueous solutions and can be predicted by the molecule's Lewis structure, which reveals its reactive sites.
Practical Tips and Best Practices
Here are some practical tips and best practices to consider when working with the NCl3 Lewis structure:
- Draw Rough Sketches: Always start by sketching a rough outline of the molecule. This helps you organize your thoughts and see the electron distribution clearly.
- Check Octet Rule Compliance: Ensure all atoms, especially the central one, comply with the octet rule. If not, adjust by redistributing electrons.
- Use Formal Charge Calculation: Calculate formal charges to find the most stable configuration. The goal is to minimize positive formal charges on more electronegative atoms.
Practical FAQ
Why is understanding the NCl3 Lewis structure important?
Understanding the NCl3 Lewis structure is important because it provides a clear depiction of how the atoms are bonded and how electrons are distributed. This understanding is crucial for predicting molecular geometry, chemical reactivity, and stability. It helps in understanding how NCl3 will interact with other molecules, particularly in industrial and environmental settings.
Common user question about practical application
What are the implications of NCl3’s Lewis structure in chemical reactions?
The NCl3 Lewis structure implies that nitrogen will act as a central atom with three chlorine atoms forming single bonds and one lone pair. This structure allows for the