If you've ever wondered about the nature of NH3, also known as ammonia, you're not alone. Many people struggle to understand whether ammonia is an acid or a base, a topic that often sparks confusion. This guide aims to clear up the mystery surrounding ammonia’s properties, offering practical, actionable advice to help you grasp this fundamental concept in chemistry. Let’s dive into the fascinating world of NH3, revealing its true identity as a base.
Understanding NH3: Is It an Acid or a Base?
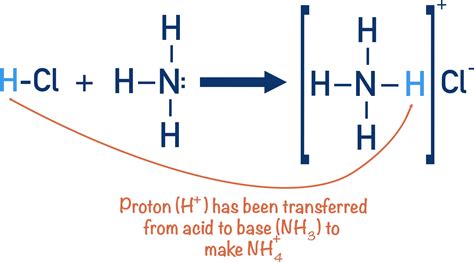
Ammonia is typically considered a base, rather than an acid. This classification may seem counterintuitive at first, especially since its properties can also resemble those of acids in certain contexts. To understand why ammonia is classified as a base, we need to delve into the basic principles of acids and bases. The classic definition of acids and bases is rooted in the Arrhenius theory, which suggests that acids are substances that donate hydrogen ions (H+) in solution, while bases donate hydroxide ions (OH-). However, in the broader scope of modern chemistry, the Bronsted-Lowry and Lewis theories expand our understanding of acids and bases, defining them in terms of proton (H+) donation and acceptance, as well as electron pair donation.
Ammonia's behavior as a base becomes clear when we examine it under these broader definitions. When NH3 dissolves in water, it accepts a hydrogen ion (H+) from water molecules, forming the ammonium ion (NH4+) and a hydroxide ion (OH-). This reaction highlights ammonia's role as a base, as it accepts protons from the water, increasing the solution's alkalinity.
Quick Reference
Quick Reference
- Immediate action item with clear benefit: Always label ammonia containers clearly to prevent misuse.
- Essential tip with step-by-step guidance: Test pH levels when ammonia is used to ensure it doesn’t cause hazardous conditions.
- Common mistake to avoid with solution: Mistakenly classify ammonia as an acid based on its acidic odor; remember it’s a base.
Why Ammonia Is a Base: Detailed Explanation
To understand why ammonia (NH3) is classified as a base, it’s crucial to explore its chemical behavior in depth. Here’s a step-by-step guide to unraveling its base nature:
Chemical Structure of Ammonia
Ammonia has a trigonal pyramidal structure with nitrogen at the center bonded to three hydrogen atoms. The nitrogen atom has a lone pair of electrons, which is a key feature for its base properties. This lone pair allows ammonia to act as a Lewis base, which is one that donates an electron pair to form a covalent bond.
Reaction with Water
When ammonia is added to water, it undergoes a reaction that highlights its base nature:
NH3 + H2O ⇌ NH4+ + OH-
In this reaction, ammonia accepts a hydrogen ion from water, forming the ammonium ion (NH4+) and increasing the concentration of hydroxide ions (OH-) in the solution, which is characteristic of a basic solution.
pH and Alkalinity
Solutions containing ammonia typically have a pH greater than 7, confirming their basic nature. The increase in OH- ions is what causes this rise in pH, making it an effective alkaline agent.
How to Use Ammonia Safely and Effectively
Using ammonia in household or industrial settings requires careful handling due to its caustic properties. Here’s how to utilize ammonia safely and effectively:
Safety First: Handling Ammonia
Before using ammonia, always ensure you are working in a well-ventilated area to avoid inhalation of its fumes. Use gloves and eye protection, and make sure to store ammonia in labeled, sealed containers away from acids and heat sources.
Applying Ammonia in Cleaning
Ammonia is widely used in cleaning solutions due to its excellent cleaning power and ability to break down grease and grime. Here’s how to mix and use it effectively:
- Dilute ammonia with water in a ratio of 1 part ammonia to 10 parts water for household cleaning.
- Test the pH of the solution after mixing to ensure it remains within a safe, basic range (pH 9-11).
- Use it to clean bathroom surfaces, windows, and floors, ensuring to rinse with clean water to remove any leftover ammonia.
Industrial Uses of Ammonia
In industrial settings, ammonia is used in a variety of applications, from refrigeration systems to manufacturing processes. Here are some steps to consider for safe usage:
- Implement ammonia in refrigeration systems only if trained and qualified to handle high-pressure and high-temperature conditions.
- Use ammonia in chemical syntheses by following specific procedures outlined by safety guidelines and manufacturers.
- Regularly monitor the integrity of storage containers and pipelines to prevent leaks and accidental releases.
Practical FAQ
Can ammonia be used as a disinfectant?
Yes, ammonia can be used as a disinfectant in diluted forms. It’s effective against many bacteria and viruses when used correctly. However, it’s crucial to use it in well-ventilated areas and avoid mixing it with other cleaning agents like bleach, which can produce toxic gases. Always follow safety instructions and use gloves and eye protection when handling ammonia for disinfection purposes.
How can I neutralize ammonia in a water solution?
To neutralize ammonia in water, you can carefully add a mild acid like vinegar (acetic acid) in small amounts to bring down the pH. However, this process should be done slowly and with caution, as it can produce heat and gas. Always perform this in a well-ventilated area and use appropriate personal protective equipment (PPE). It’s important to avoid creating dangerous chemical reactions by adding acids to ammonia.
This detailed guide offers a comprehensive understanding of ammonia’s nature as a base, its chemical interactions, and its safe use in various applications. By following these practical tips and procedures, you can effectively utilize ammonia while minimizing risks to yourself and your environment.