Nitric acid, with the chemical formula HNO3, is a highly corrosive and toxic strong acid that is commonly used in the production of fertilizers, explosives, and as a laboratory reagent. Understanding its molecular structure is crucial for grasping its chemical properties and reactivity. The Lewis structure of a molecule is a simplified representation that shows the arrangement of electrons in the molecule, helping to identify the bonding and lone pairs around each atom.
Nitric Acid Molecular Structure
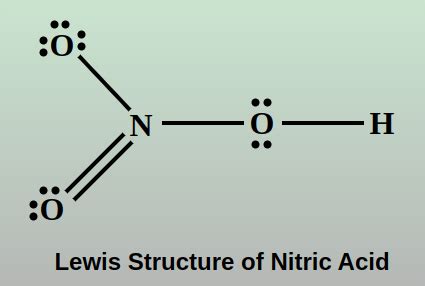
The nitric acid molecule consists of one hydrogen atom, one nitrogen atom, and three oxygen atoms. To draw the Lewis structure of HNO3, we start by counting the total number of valence electrons. Hydrogen has 1 valence electron, nitrogen has 5, and each oxygen has 6, giving a total of 1 + 5 + (3*6) = 24 valence electrons. Next, we connect the atoms with single bonds, which uses 6 electrons, leaving 18 electrons to distribute as lone pairs around the atoms to satisfy the octet rule for each atom, except for hydrogen, which needs only 2 electrons to fill its 1s orbital.
Constructing the Lewis Structure
The construction of the Lewis structure for nitric acid involves several steps. First, the nitrogen atom is typically placed at the center because it can form more bonds than hydrogen. The hydrogen atom is then bonded to one of the oxygen atoms or directly to the nitrogen, but in the case of nitric acid, the most stable and commonly depicted structure has the hydrogen bonded to one of the oxygens. Each oxygen atom is then connected to the nitrogen atom with a single bond. This initial structure accounts for 6 electrons in bonding pairs. The remaining 18 electrons are distributed as lone pairs: 3 lone pairs (6 electrons) on each of the two oxygen atoms that are singly bonded to nitrogen and 1 lone pair (2 electrons) on the nitrogen atom, and the oxygen atom bonded to hydrogen will have 2 lone pairs (4 electrons) on it as well, but to satisfy the octet rule and the observed chemistry of nitric acid, one of the bonds between nitrogen and an oxygen must be a double bond, which consumes 2 more electrons and adjusts the lone pair distribution accordingly.
| Atom | Valence Electrons | Bonding Electrons | Lone Pair Electrons |
|---|---|---|---|
| Hydrogen | 1 | 2 (shared) | 0 |
| Nitrogen | 5 | 4 (shared in single and double bonds) | 2 (1 lone pair after satisfying octet) |
| Oxygen (double bonded) | 6 | 4 (2 in single bond, 2 in double bond, 4 shared) | 4 (2 lone pairs) |
| Oxygen (single bonded to N, not to H) | 6 | 2 (shared in single bond) | 6 (3 lone pairs) |
| Oxygen (single bonded to H and N) | 6 | 4 (2 in single bond to H, 2 in single bond to N) | 4 (2 lone pairs) |

Key Points

Key Points
- Nitric acid (HNO3) has a complex molecular structure that involves a nitrogen atom bonded to three oxygen atoms, with one of these bonds being a double bond to satisfy the octet rule for nitrogen.
- The Lewis structure is a critical tool for visualizing the molecular structure and electron distribution, which influences the chemical properties of the molecule.
- Understanding the bonding and lone pairs in the Lewis structure of nitric acid helps in predicting its reactivity and interactions with other molecules.
- The structure involves a balance between satisfying the octet rule for each atom (except hydrogen, which follows the duet rule) and minimizing formal charges on the atoms.
- The double bond in the nitric acid molecule plays a significant role in its stability and chemical reactivity, contributing to its properties as a strong acid and an oxidizing agent.
Implications of the Lewis Structure
The Lewis structure of nitric acid has significant implications for its chemical properties and behavior. The presence of the double bond between nitrogen and oxygen, along with the distribution of lone pairs, influences the molecule’s polarity and its ability to participate in chemical reactions. The structure is essential for understanding the acid’s strength, its oxidizing properties, and its role in various industrial and laboratory applications.
Nitric acid's strong acidity can be partly attributed to the stability of its conjugate base (NO3-) and the facility with which the hydrogen atom can be released as a proton (H+). This aspect, combined with its oxidizing power due to the presence of the double bond and the high electronegativity of the oxygen atoms, makes nitric acid a versatile reagent in organic and inorganic chemistry.
Conclusion and Future Perspectives
In conclusion, the Lewis structure of nitric acid provides a fundamental framework for understanding its chemical properties and reactivity. The arrangement of electrons, the types of bonds, and the distribution of lone pairs around each atom are crucial for predicting how nitric acid will behave in different chemical environments. As research continues to explore new applications of nitric acid and its derivatives, a deep understanding of its molecular structure will remain essential for advancing our knowledge in fields such as chemistry, materials science, and environmental science.
What is the significance of the double bond in the nitric acid Lewis structure?
+The double bond between nitrogen and one of the oxygen atoms is crucial for the stability and reactivity of the nitric acid molecule. It influences the molecule’s polarity and its ability to act as a strong acid and an oxidizing agent.
How does the Lewis structure of nitric acid relate to its properties as a strong acid?
+The Lewis structure helps explain the stability of the nitrate ion (NO3-), the conjugate base of nitric acid, which is a key factor in its acidity. The ease with which the hydrogen atom can be released as a proton is influenced by the molecular structure.
What role does the distribution of lone pairs play in the chemical properties of nitric acid?
+The distribution of lone pairs around the nitrogen and oxygen atoms affects the molecule’s polarity and its ability to form hydrogen bonds, which are important for its solubility in water and its interactions with other molecules.


