Lithium is an element that holds significant importance in both chemistry and various technological applications. One intriguing aspect of lithium is its atomic structure, specifically its proton count. Understanding the proton count in lithium can provide practical insights into its chemical behavior and practical applications.
The Proton Count in Lithium
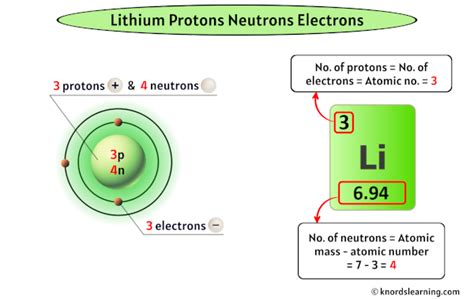
Lithium, with the atomic number 3, is the lightest metal and the lightest solid element in general. It is comprised of three fundamental particles: protons, neutrons, and electrons. A key characteristic of any element is its proton count, which defines its atomic number and determines its position on the periodic table. For lithium, this count is straightforward and fundamental to its identity: lithium has three protons. This proton count governs the element’s chemical properties and behavior.The Relevance of Lithium’s Proton Count
Understanding the proton count in lithium is not just a matter of academic interest; it has significant practical implications. For example, lithium’s proton count affects its role in nuclear chemistry and its use in nuclear reactors as a neutron absorber. Furthermore, in chemistry, the three protons of lithium ensure that its valency is +1, influencing its reactivity and how it forms compounds.Lithium in Technological Applications
The proton count of lithium is a fundamental element in various technological advancements. One prominent area is lithium-ion batteries. These batteries rely on lithium’s unique properties to store and release energy efficiently. The stable proton count ensures consistent performance, which is critical for devices ranging from smartphones to electric vehicles. Additionally, in the medical field, lithium salts are used in psychiatric treatment, highlighting the importance of understanding this element’s atomic structure.Key Insights
Key Insights
- Primary insight with practical relevance: Lithium’s proton count of three is crucial in defining its chemical properties and technological applications.
- Technical consideration with clear application: The consistent proton count of lithium ensures reliability in applications like lithium-ion batteries.
- Actionable recommendation: For scientists and engineers, a fundamental understanding of lithium’s proton count can lead to improved technological innovations.
Scientific Importance of Proton Count
In scientific research, the atomic structure of elements such as lithium informs numerous studies and applications. The proton count determines the nucleus’s charge, which directly influences the atom’s chemical behavior. For instance, lithium’s +1 valency plays a pivotal role in forming ionic compounds, which are foundational in many chemical reactions and processes.FAQ Section
What makes lithium special?
Lithium’s special attributes stem from its low atomic mass and unique proton count of three, making it highly reactive and essential in various technological and medical applications.
How does lithium’s proton count affect battery technology?
Lithium’s proton count and its resulting +1 valency are essential for the consistent performance and energy storage efficiency of lithium-ion batteries.
Understanding the fundamental aspects of lithium’s atomic structure, particularly its proton count, is pivotal for advancing scientific research and technological development. This knowledge provides a basis for the practical application of lithium in various fields, ensuring continued progress in innovation and efficiency.