The pursuit of mastering the O3 Lewis structure represents a critical journey for chemistry students and professionals alike. Understanding this structure is fundamental in grasping concepts like molecular geometry, bond angles, and formal charges. The O3 molecule, or ozone, exemplifies unique aspects of molecular bonding, making it a perfect subject for in-depth analysis and practical understanding.
Introduction
The O3 molecule, composed of three oxygen atoms, offers a fascinating insight into molecular structure and bonding. Correctly drawing its Lewis structure is not merely an academic exercise but a gateway to comprehending more complex chemical systems. This article will guide you through the step-by-step process, providing practical insights and evidence-based knowledge to unlock the mystery behind the O3 Lewis structure.
Key Insights
- The O3 molecule exhibits resonance structures, a fundamental concept for understanding molecular bonding.
- Correctly calculating and distributing valence electrons is crucial for accurate Lewis structure drawing.
- Utilizing the octet rule and understanding formal charge calculations enhances the accuracy and practical application.
Understanding the Basics
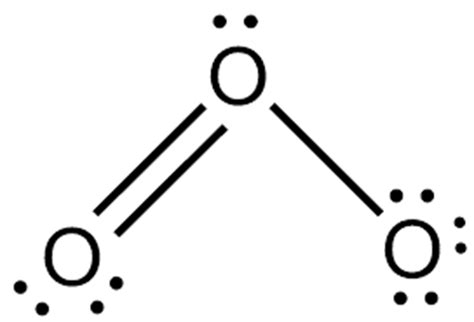
To draw the O3 Lewis structure, it’s essential to grasp the basics of valence electrons and molecular geometry. Ozone consists of three oxygen atoms, each with six valence electrons. Drawing an accurate Lewis structure requires us to correctly distribute these electrons to satisfy the octet rule for each atom. Start by placing the atoms in a way that forms double bonds. One oxygen atom will be the central atom, while the other two will form a double bond with it, each oxygen on the outer ends sharing a single bond with the central oxygen. This arrangement allows us to begin the process of filling octets around each atom.
Resonance Structures and Formal Charges
O3 exhibits resonance structures, a concept where multiple valid Lewis structures can represent a molecule due to the delocalization of electrons. In the O3 molecule, the double bond can shift between the two outer oxygen atoms, leading to two primary resonance forms. Each resonance structure contributes to the overall stability of the molecule, and it’s crucial to understand the implications of these structures. Calculating formal charges for each atom in potential resonance forms is vital. Formal charge helps determine the most stable resonance structure and ensures that the electron distribution adheres to chemical principles.
What is the significance of resonance in O3?
Resonance in O3 helps explain its stability and molecular geometry by showing how electrons can be shared between atoms, providing a more accurate depiction of its true structure than any single Lewis structure could.
Why is the octet rule important for O3?
The octet rule ensures that each atom in O3 achieves a stable electron configuration by filling its outer shell with eight electrons, which is fundamental for the molecule's stability and reactivity.
Drawing the O3 Lewis structure is a practical application of several key principles in chemistry. By understanding the basics of valence electrons, employing resonance structures, and applying the octet rule and formal charges, one can accurately represent the O3 molecule. This exercise not only enhances one’s ability to draw complex Lewis structures but also deepens the understanding of molecular bonding and stability.