Understanding Solubility Curves: Expert Perspective and Practical Insights
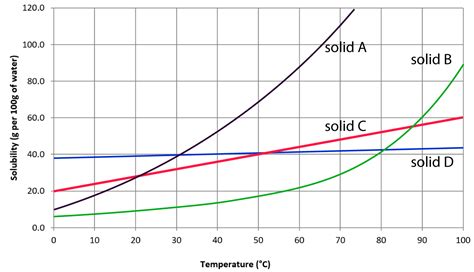
When delving into the realm of solubility, the term “saturated” holds significant importance. A solubility curve is a graphical representation illustrating the maximum amount of solute that can dissolve in a solvent at specific temperatures. The point at which no more solute can dissolve in a given solvent is termed as saturated. This condition is crucial for numerous scientific, industrial, and even everyday applications.
The context of saturation becomes pivotal when discussing how solubility changes with temperature. The practical implications of this include everything from creating solutions for pharmaceutical compounds to determining the right ratios in chemical reactions and industrial processes.
Key Insights
Key Insights
- A saturated solution indicates the maximum solubility of a substance at a specific temperature.
- Understanding solubility curves helps in predicting solute behavior in various conditions.
- Knowledge of saturated solutions can guide decisions in chemical engineering, pharmaceuticals, and food science.
When exploring solubility curves, one should pay close attention to how the slope of the curve affects the solubility of different substances. Generally, the steeper the slope, the more solubility increases with temperature. This characteristic is critical when dealing with solutes like potassium nitrate versus substances like sodium chloride.
Moreover, the interaction between temperature and solubility has real-world applications. For instance, when preparing a concentrated sugar solution for candy-making, understanding the saturation point at different temperatures can make the difference between a perfect batch and a poorly crystallized one.
Two analysis sections with
headings follow to dive deeper into the technical aspects and practical implications of solubility curves.
Analyzing Solubility Curves and Temperature Effects
A thorough understanding of solubility curves is instrumental in predicting how solutes behave under varying conditions. An upward-sloping solubility curve indicates that as temperature increases, so does the amount of solute that can dissolve in the solvent. This principle is vividly illustrated when looking at substances such as potassium nitrate.
For example, if we take potassium nitrate, its solubility increases significantly with temperature; at 20°C, it dissolves to about 32 grams per 100 grams of water, and this amount escalates to around 64 grams at 80°C. Such knowledge is indispensable in industries where precise control over solubility is crucial, such as in the crystallization process in pharmaceuticals.
Conversely, a solubility curve with a shallow slope, like that for sodium chloride, shows a more gradual increase in solubility with temperature. Here, at 20°C, the solubility is approximately 36 grams per 100 grams of water, rising to 38.8 grams at 100°C. The difference in slopes between these two substances exemplifies the diversity in solute behavior, which can profoundly impact process designs in chemical manufacturing.
FAQ Section
How do solubility curves aid in pharmaceutical manufacturing?
Solubility curves provide crucial information on the maximum amount of solute that can be dissolved in a solvent at specific temperatures. This information helps in designing formulations where precise dosing and dissolution rates are critical for efficacy and patient safety.
What happens when a solution exceeds its saturation point?
When a solution exceeds its saturation point, it becomes supersaturated, meaning that it contains more solute than the solubility curve indicates it should hold at that temperature. This can result in the formation of undissolved crystals as the solution tries to reach equilibrium.
Understanding solubility curves and the concept of saturation is not just an academic exercise but a practical necessity across numerous fields. Whether it’s optimizing reaction conditions in the lab or ensuring consistent product quality in industrial processes, the insights derived from these curves are invaluable.