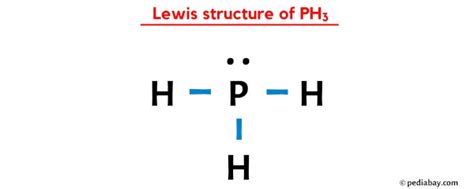
Understanding the Ph3 (phosphine) Lewis structure can significantly enhance your grasp of molecular bonding and geometry in organic and inorganic chemistry. Ph3, often used in coordination chemistry, provides a fascinating case study for analyzing the role of lone pairs and the octet rule. This article delves into the nuances of constructing the Ph3 Lewis structure, ensuring a robust comprehension of electron distribution and molecular configuration.
The Basics of Lewis Structures
Lewis structures are fundamental tools in chemistry for representing valence electrons and molecular bonding. For Ph3, we must consider the phosphorus atom central with three hydrogen atoms bonded via single covalent bonds and a lone pair of electrons on the phosphorus. Each hydrogen atom requires two electrons to achieve a full outer shell, while phosphorus aims to complete its octet.
Constructing the Ph3 Lewis Structure
When constructing the Ph3 Lewis structure, begin by placing the phosphorus atom at the center, surrounded by three hydrogen atoms. Connect each hydrogen atom with a single bond to phosphorus, accounting for the eight electrons shared in these bonds. Phosphorus has five valence electrons, and each hydrogen requires two electrons. After satisfying the hydrogen atoms, two electrons remain on phosphorus, forming a lone pair. This lone pair is crucial for understanding Ph3’s geometry and chemical behavior.
Key Insights
- Primary insight with practical relevance: The Ph3 Lewis structure illustrates the importance of lone pairs in molecular geometry.
- Technical consideration with clear application: Understanding Ph3’s Lewis structure can guide predictions about reactivity in chemical reactions.
- Actionable recommendation: Always consider lone pairs when determining molecular shapes and reactivity.
Exploring Electron Distribution
The electron distribution in the Ph3 molecule adheres to the octet rule, although phosphorus can expand its octet due to its ability to accommodate more than eight electrons. The three hydrogen atoms, bonded via single covalent bonds, ensure that phosphorus shares its electrons efficiently, completing the octet for each hydrogen and retaining a lone pair. This configuration leads to a trigonal pyramidal molecular geometry, as the lone pair exerts a greater repulsive force than bonding pairs, affecting the spatial arrangement.
Octet Rule and Exceptions
Phosphorus’s ability to exceed the traditional octet rule exemplifies its higher valence electron capacity due to its position in the periodic table. Ph3’s structure showcases a critical understanding of the octet rule’s limitations, especially for elements beyond the second period. While most second-period elements adhere strictly to the octet rule, third-period elements like phosphorus can form expanded octets, crucial for grasping advanced chemical concepts.
Why does the lone pair affect molecular geometry?
The lone pair on phosphorus repels bonding pairs more strongly due to closer electron density proximity, causing a distortion in the ideal trigonal planar arrangement, resulting in a trigonal pyramidal shape.
Can Ph3 form coordination compounds?
Yes, Ph3 can act as a ligand in coordination compounds due to the lone pair on phosphorus, which can donate electron density to a metal center, forming coordinate covalent bonds.
In conclusion, mastering the Ph3 Lewis structure requires attention to electron distribution, the role of lone pairs, and the octet rule’s nuances. Understanding these elements not only clarifies the molecular geometry but also enhances your predictive capabilities in various chemical contexts. This detailed approach ensures a comprehensive and practical understanding of Ph3, empowering you with key insights for advanced studies in chemistry.