If you're struggling to find the right formula for potassium bromide, this guide will walk you through everything you need to know in a straightforward and practical way. Whether you're a chemistry enthusiast, a student, or someone dealing with specific health-related needs, understanding the correct potassium bromide formula is crucial for accurate and effective application. Let's dive right in to address your immediate needs and equip you with actionable advice and real-world solutions.
Understanding the Importance of the Potassium Bromide Formula
Potassium bromide (KBr) is a compound commonly used in medical treatments, particularly as a sedative and anticonvulsant. It is also used in various industrial applications and research settings. Knowing the correct formula ensures that you can prepare solutions accurately, whether for laboratory experiments or medical uses. Incorrect usage can lead to ineffective results or, in some cases, serious health risks.
Quick Reference
Quick Reference
- Immediate action item with clear benefit: Always verify the molar mass of potassium bromide to ensure the right amount for your specific needs.
- Essential tip with step-by-step guidance: To prepare a potassium bromide solution, start by weighing out your precise amount of KBr, then dissolve it in the correct volume of water.
- Common mistake to avoid with solution: Do not confuse potassium bromide (KBr) with potassium bromate (KBrO3) as they have different applications and health implications.
How to Prepare a Potassium Bromide Solution
Preparing a potassium bromide solution requires attention to detail to ensure accuracy and safety. Here’s a step-by-step guide to preparing a potassium bromide solution:
Step 1: Understanding the Formula
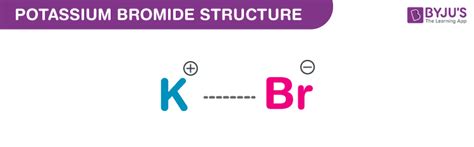
Potassium bromide (KBr) is composed of one potassium (K) atom and one bromine (Br) atom. The molar mass of KBr is approximately 119 g/mol. Understanding this allows you to accurately measure the compound when preparing solutions. The correct formula ensures that you use the right amount of KBr for your needs, be it for laboratory work or medical application.
Step 2: Gathering Your Materials
To prepare a KBr solution, you’ll need:
- Potassium bromide (KBr) crystals
- Distilled water
- A digital weighing scale
- A beaker or flask
- A stirring rod or magnetic stirrer
Step 3: Measuring the Correct Amount
First, you need to determine the amount of KBr required for your solution. Suppose you need to prepare a 0.1 M (molar) solution of KBr in 1 liter of water. Using the molar mass of KBr (119 g/mol), you can calculate the weight needed as follows:
0.1 mol/L * 1 liter * 119 g/mol = 11.9 grams of KBr
Weigh 11.9 grams of KBr crystals accurately using a digital scale. Precision here is key to ensure your solution's efficacy.
Step 4: Dissolving the Potassium Bromide
Place the measured KBr crystals in a beaker or flask. Begin to dissolve the KBr in a smaller volume of distilled water, around half a liter, to avoid rapid dissolution which can cause splattering. Stir the mixture gently to speed up the dissolution process.
Important: Make sure to use distilled or deionized water to avoid contamination from impurities that could affect the solution’s properties.
Step 5: Making Up to Volume
Once the KBr is fully dissolved, carefully transfer the solution to a 1-liter volumetric flask. Add more distilled water to the flask until the bottom of the meniscus is at the calibration mark. This ensures that the final volume is exactly 1 liter, providing an accurate 0.1 M solution.
Practical Applications of Potassium Bromide
Potassium bromide has multiple practical applications, making it essential to understand its preparation and handling correctly.
Medical Use
KBr is primarily used in medical treatments as a sedative and anticonvulsant. It works by depressing the central nervous system, which can be particularly useful in managing conditions such as epilepsy and anxiety.
Industrial Applications
In industry, KBr is used as a chemical reagent in various reactions, including the creation of photographic chemicals and in electroplating processes. Its role as a bromine source makes it indispensable in certain specialized applications.
Laboratory Use
For laboratory work, KBr is often used in calibration standards and in analytical chemistry. Its well-defined properties make it a reliable compound for various standard solutions and tests.
Practical FAQ
How do I store potassium bromide safely?
Storage Tips: Store potassium bromide in a cool, dry place, away from sources of heat and moisture. Keep the container tightly sealed to prevent contamination and degradation. Avoid exposure to air and light, which can degrade the compound over time.
If you need to store it for an extended period, ensure the storage area is free from any substances that could react with KBr, as this could compromise the purity and effectiveness of the compound.
What are the safety precautions when handling potassium bromide?
Safety Measures: When handling potassium bromide, it is essential to wear appropriate personal protective equipment (PPE) such as gloves, safety goggles, and a lab coat. Potassium bromide can be irritating to the skin and eyes, so taking these precautions can prevent accidental exposure.
Ensure good ventilation in the work area to avoid inhalation of any dust that may become airborne. In case of skin contact, wash the affected area thoroughly with water. If you experience any discomfort or irritation, seek medical attention immediately.
Can I use regular tap water to prepare a potassium bromide solution?
No: Do not use tap water to prepare a potassium bromide solution. Tap water contains various impurities and minerals that can interfere with the purity and efficacy of the solution. Instead, always use distilled or deionized water to ensure the highest quality of your KBr solution.
Understanding and correctly using the potassium bromide formula can make a significant difference in both its effectiveness and safety. By following these guidelines and tips, you can confidently prepare potassium bromide solutions and utilize its many applications across various fields. Always remember to handle this compound with care and precision to achieve the best results.