The Specific Heat of Lead: Uncover Its Mysteries
Understanding the specific heat of lead is crucial for many practical applications, especially in industrial and scientific fields. Specific heat is the amount of heat required to change the temperature of a unit mass of a substance by one degree Celsius. Lead, a dense metal with numerous applications from ammunition to electrical components, has a specific heat that plays a role in these uses. This guide aims to unpack the mysteries of lead’s specific heat, addressing your practical needs with step-by-step guidance, real-world examples, and expert tips.
Understanding lead’s specific heat can seem daunting at first glance, but this guide will break down the process into manageable parts. Whether you’re an engineer designing cooling systems, a chemist studying thermal properties, or just curious about this heavy metal’s thermal characteristics, this guide will serve as your comprehensive resource. With actionable advice, practical solutions, and detailed how-to sections, you’ll not only understand the concept but also apply it effectively in your field of work.
Why Knowing the Specific Heat of Lead Matters
Knowing the specific heat of lead is vital for optimizing thermal processes. It helps in designing better cooling systems, predicting thermal behavior in lead-based applications, and ensuring the safety of structures and systems where lead is used. Misunderstanding or neglecting the specific heat can lead to overheating, failure of materials, and even safety hazards. This guide will walk you through the specifics, ensuring that you can confidently handle lead-related thermal challenges.
Quick Reference
Quick Reference
- Immediate action item: Calculate the specific heat of lead to determine its thermal properties.
- Essential tip: Use the formula q = mcΔT where q is heat, m is mass, c is specific heat capacity, and ΔT is temperature change.
- Common mistake to avoid: Confusing specific heat with thermal conductivity; remember, specific heat measures heat required per degree change, while thermal conductivity measures heat transfer rate.
How to Measure the Specific Heat of Lead
To accurately measure the specific heat of lead, you’ll need to follow a systematic approach. Here’s a detailed step-by-step guide to get precise results:
Step 1: Gather Your Materials
Before starting, ensure you have the following materials and equipment:
- A lead sample of known mass
- A thermometer with a fine range
- A calorimeter or a heat-exchange setup
- Water or another liquid for the calorimetry process
- A heating source
- Safety gear (gloves, goggles)
Step 2: Set Up Your Experiment
Place the lead sample in a calorimeter or setup that allows for controlled heating and temperature measurement. Make sure the sample is isolated to avoid heat loss to the surroundings.
Step 3: Heating the Lead Sample
Use a controlled heating source to incrementally heat the lead sample. Record the temperature of the lead at regular intervals to observe its thermal response. Ensure that the heating is gradual to prevent any abrupt changes in temperature.
Step 4: Measuring Temperature Changes
Simultaneously, keep track of the temperature of the water or liquid medium used in your calorimeter. This will help in calculating the heat exchanged between the lead sample and the medium.
Step 5: Calculating the Specific Heat
Using the formula q = mcΔT, where q is the heat absorbed by the lead sample, m is the mass of the lead, and ΔT is the temperature change, calculate the specific heat capacity of lead. Repeat the experiment multiple times to ensure accuracy.
Step 6: Analyze and Compare Results
Compare your calculated specific heat values with standard references. Ensure your results are consistent across multiple trials to validate your measurements.
Practical Application of Lead’s Specific Heat
Understanding lead’s specific heat is just the first step; applying this knowledge is crucial for practical scenarios:
Cooling Systems Design
When designing cooling systems for lead components, knowing the specific heat allows for accurate estimation of the amount of heat lead can absorb without a significant temperature rise. This is especially important in manufacturing processes where temperature control is critical.
Safety Measures
In industries where lead is used extensively, understanding its specific heat helps in predicting temperature changes during operations. This knowledge can inform safety measures, such as protective equipment and cooling strategies to prevent overheating and related hazards.
Thermal Management in Electronics
Lead is sometimes used in electronic components, and its thermal properties can influence the design and functionality of circuits. Accurate knowledge of its specific heat ensures that thermal management systems are effective and prevent damage from thermal stress.
Practical FAQ
How does lead’s specific heat compare to other metals?
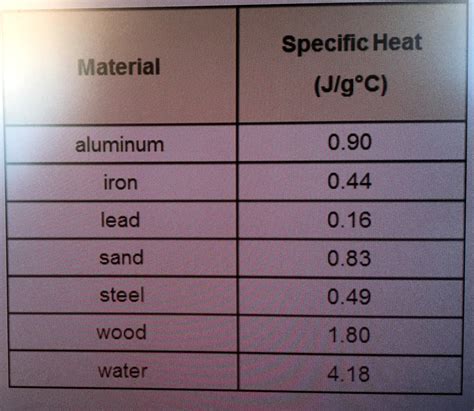
Lead has a specific heat of approximately 0.16 J/g°C, which is relatively low compared to metals like copper (0.39 J/g°C) or aluminum (0.9 J/g°C). This means that lead absorbs and retains less heat per unit mass than these other metals. When designing systems involving multiple metals, this difference in specific heat needs to be considered to ensure balanced thermal performance.
Common Mistakes and How to Avoid Them
Understanding lead’s specific heat comes with a few common pitfalls that can lead to incorrect results or misunderstandings:
- Mistake: Failing to account for heat loss to the environment. Solution: Conduct your experiments in an insulated environment to minimize heat loss, ensuring more accurate measurements.
- Mistake: Using incorrect units for mass or temperature. Solution: Always use SI units (kilograms for mass and degrees Celsius for temperature) to ensure consistency and accuracy in your calculations.
- Mistake: Not calibrating your thermometer and equipment before starting the experiment. Solution: Calibrate all your measuring instruments to the standard temperature before beginning your experiment to ensure precise readings.
By avoiding these common mistakes and following the detailed steps provided, you can confidently measure and apply the specific heat of lead in your practical applications.
Through this guide, you now possess the knowledge and practical steps necessary to understand and utilize the specific heat of lead effectively. Whether you are working on industrial projects, scientific research, or simply enhancing your understanding of this important metal, this guide serves as a comprehensive resource to unlock the mysteries of lead’s specific heat.