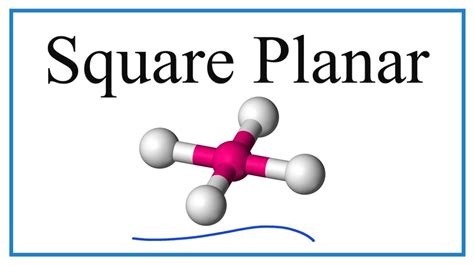
Imagine diving into the depths of inorganic chemistry, a place where molecules adopt unique shapes and behaviors, driven by the fundamental forces governing electron interactions. Here, we delve into the world of square planar complexes, a category of coordination compounds that exhibit remarkable properties, poised to unlock new chapters in chemical exploration and applications.
Key insights box:
Key Insights
- Square planar complexes often feature transition metals in the d8 configuration, showcasing a unique geometry that enables versatile coordination chemistry.
- Their electronic structure is instrumental in catalysis, affecting the energy levels and stabilization of reaction intermediates.
- An actionable recommendation would be to explore the use of square planar complexes in homogeneous catalysis to improve efficiency and selectivity in chemical reactions.
Square planar complexes have emerged as pivotal entities in coordination chemistry due to their distinct geometry and electronic configuration. Typically, these complexes are composed of a central metal ion, surrounded by four ligands in a planar arrangement. The predominant metals involved are from the transition series, especially those in the d8 electron configuration, such as platinum(II) and palladium(II). This geometry, while stable, offers unique opportunities for chemical interactions that can be exploited in various industrial and research applications.
One fascinating aspect of square planar complexes is their role in catalysis. These complexes stabilize transition states and intermediates through coordination interactions, facilitating chemical reactions that might otherwise be prohibitively slow or energetically unfavorable. Their planar structure ensures optimal overlap of metal d-orbitals with ligand orbitals, which is crucial for effective electron transfer during catalysis. This is exemplified by the use of square planar platinum complexes in homogeneous catalysis, significantly impacting the pharmaceutical industry for the synthesis of complex organic molecules.
The stability and reactivity of square planar complexes also lie at the intersection of electronic structure and coordination environment. The d8 configuration allows for significant ligand-field splitting, which in turn affects the electronic transitions and overall reactivity of the complex. The ability to fine-tune these properties by varying ligands is a powerful tool for chemists, enabling the development of catalysts tailored for specific reactions. For instance, the introduction of electron-donating or withdrawing ligands can modulate the electronic environment, thus influencing the complex’s catalytic efficiency and selectivity.
The strategic manipulation of square planar complexes can drive advancements in various chemical processes. Their application extends to areas such as organometallic synthesis, where their unique coordination capabilities enable the formation of intricate molecular architectures. Additionally, their involvement in transition metal-catalyzed cross-coupling reactions underscores their importance in creating new carbon-carbon bonds essential for organic synthesis.
FAQ section:
What are the primary applications of square planar complexes?
Square planar complexes are primarily used in homogeneous catalysis, where their unique geometry and electronic properties enable effective stabilization of reaction intermediates. They are also crucial in organometallic synthesis for constructing complex molecular structures.
How does the geometry of square planar complexes influence their catalytic properties?
The planar geometry of square planar complexes allows for optimal orbital overlap between the metal center and ligands, facilitating efficient electron transfer during catalytic cycles. This geometry also influences the stability and reactivity of transition states, enhancing catalytic activity.
In summary, square planar complexes offer a compelling platform for advancing chemical research and industrial applications. Their distinctive geometry, combined with the rich interplay of electronic structure and coordination environment, positions them as critical players in the realm of coordination chemistry and catalysis. With continued exploration and innovation, these complexes are set to catalyze new discoveries and breakthroughs in chemical science.