Valence Electrons Aluminum Unveiled: Surprising Secrets!
Understanding the concept of valence electrons is fundamental in the world of chemistry, especially when it comes to elements like aluminum. Valence electrons determine how an element interacts with others to form chemical bonds and compounds. While the principles might seem straightforward, diving deep into the details unveils surprising secrets about aluminum’s behavior in various chemical reactions. In this guide, we will walk you through everything you need to know about valence electrons in aluminum, providing actionable advice, practical examples, and addressing common pain points you might encounter.
Whether you're a student trying to grasp this concept for a class project or a professional chemist seeking to refine your knowledge, this guide will offer you clear, step-by-step guidance that’s easy to implement. We’ll start by addressing some prevalent problems and then move to provide solutions and practical insights.
Problem-Solution Opening: The Fundamentals of Aluminum’s Valence Electrons
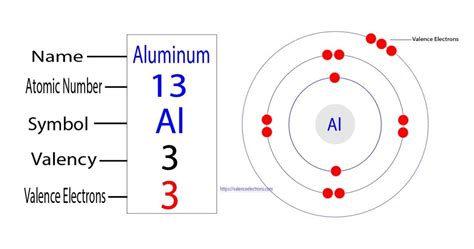
Many learners often struggle to fully comprehend the concept of valence electrons, particularly when it comes to specific elements like aluminum. The periodic table tells us that aluminum has the atomic number 13. Its electron configuration is 1s2 2s2 2p6 3s2 3p1. The part that gets tricky is understanding which electrons are considered valence electrons. Valence electrons are the outermost electrons of an atom, and they play a crucial role in chemical bonding and reactivity. For aluminum, these are the electrons in the 3s and 3p orbitals. However, grasping this alone is not enough; you need to understand how these electrons interact in real-world applications.
This guide is designed to demystify the behavior of aluminum’s valence electrons, addressing common misconceptions and providing actionable insights. Let’s get started by highlighting some key points that will make the journey more manageable.
Quick Reference
- Immediate action item with clear benefit: Start with determining the electron configuration to identify the valence electrons accurately. This will give you a clear picture of what you’re working with.
- Essential tip with step-by-step guidance: Use the Aufbau principle, which states that electrons fill the lowest energy orbitals first, to map out the electron configuration of aluminum.
- Common mistake to avoid with solution: Mistaking core electrons for valence electrons. Remember that only the outermost electrons are considered valence electrons; understanding this will prevent errors in your analysis.
Identifying Valence Electrons in Aluminum: A Detailed Walkthrough
To understand aluminum’s valence electrons, we need to dive into its electron configuration. This is crucial because it highlights the positions of the electrons in various orbitals. Aluminum’s atomic number 13 means it has 13 electrons. The distribution of these electrons in energy levels follows a specific order. Here’s a detailed breakdown:
- Step 1: Write the Electron Configuration
- Step 2: Identify the Valence Shell
- Step 3: Count the Valence Electrons
The electron configuration of aluminum is 1s2 2s2 2p6 3s2 3p1. To identify valence electrons, focus on the outermost shell.
In this configuration, the outermost shell is the third energy level, or n=3 shell. The electrons in this shell are your valence electrons.
In aluminum’s case, the 3s orbital contains 2 electrons and the 3p orbital contains 1 electron. Therefore, there are a total of 3 valence electrons in aluminum.
These electrons are critical as they are involved in the formation of bonds. For example, when aluminum forms compounds, it tends to lose these three electrons to achieve a stable electron configuration similar to that of a noble gas.
Real-World Application: How Valence Electrons Affect Chemical Reactions
Now that you understand where to find aluminum’s valence electrons, let’s delve into how these electrons influence chemical reactions. This understanding is key for both theoretical studies and practical applications.
Aluminum’s valence electrons make it highly reactive, especially in the context of oxidation reactions. When aluminum interacts with oxygen, it readily loses its three valence electrons to form aluminum oxide (Al2O3). This reaction is critical in various industries, from the manufacturing of lightweight alloys to rust prevention.
Practical Example: Aluminum in Everyday Life
To make this concept more tangible, let’s look at a practical example: aluminum foil. Aluminum foil is widely used for cooking, wrapping food, and even in industrial processes like electrical connections. The reactivity of aluminum’s valence electrons is what keeps aluminum foil from oxidizing easily in a kitchen environment. However, it does form a thin layer of aluminum oxide on its surface, which protects the metal beneath from further oxidation. Understanding this protective layer can help you appreciate the practical implications of valence electron behavior.
Practical FAQ
Why does aluminum form a protective oxide layer?
Aluminum readily loses its three valence electrons to form aluminum oxide (Al2O3). This reaction creates a thin, protective layer of aluminum oxide on the surface. This layer acts as a barrier that prevents further oxidation of the underlying metal. This protective oxide layer is why aluminum does not rust like iron, making it an excellent material for various applications such as foil and packaging.
How can I determine the number of valence electrons in other elements?
To determine the number of valence electrons in any element, follow these steps:
- Step 1: Write the element’s electron configuration.
- Step 2: Identify the highest energy level (n=3 for aluminum).
- Step 3: Count the electrons in the outermost shell.
For example, for silicon (atomic number 14), the electron configuration is 1s2 2s2 2p6 3s2 3p2. The valence electrons are in the 3rd energy level (3s2 3p2), so silicon has 4 valence electrons.
Tips and Best Practices for Mastering Valence Electrons
Mastering the concept of valence electrons will enhance your understanding of chemistry and enable you to predict chemical behavior more accurately. Here are some tips and best practices to help you:
- Practice Regularly: Regularly practice writing electron configurations and identifying valence electrons for different elements. This practice will reinforce the concepts and improve your skills.
- Use Visual Aids: Diagrams and charts can be very helpful. Visual aids like orbital diagrams can clarify the distribution of electrons in different shells.
- Engage with Real-World Examples: Apply theoretical knowledge to real-world examples. This will solidify your understanding and demonstrate the practical applications of valence electrons.
- Review Periodic Trends: Familiarize yourself with trends in the periodic table. Groups and periods provide clues about an element’s electron configuration and reactivity.
By incorporating these tips into your study routine, you will find yourself more adept at identifying valence electrons and understanding their roles in chemical reactions.
In conclusion, understanding the valence electrons in aluminum opens up a deeper comprehension of its chemical properties and behavior. By breaking down the concept into manageable steps and using practical examples, we hope this guide has provided you with the knowledge and tools to master this fundamental chemistry topic. Keep practicing and exploring, and you’ll continue to uncover the surprising secrets of elements like aluminum!