Are you curious about the fundamental components that make up the diverse world of lipids? Lipids are crucial for numerous biological functions, playing key roles in cell structure, energy storage, and signaling. Understanding their building blocks, or monomers, is essential for anyone delving into biochemistry, nutrition, or even general biology. In this guide, we will unravel the core components of lipids, giving you step-by-step guidance, practical examples, and actionable tips to grasp these essential concepts thoroughly.
Understanding Lipids: The First Step
Lipids are a diverse group of naturally occurring molecules that include fats, oils, waxes, phospholipids, and steroids. They are characterized by their hydrophobic (water-repelling) nature. While carbohydrates and proteins are soluble in water, lipids are not, making them insoluble in water and soluble in non-polar solvents. This fundamental characteristic plays a key role in the structure and function of cell membranes.
Why Do We Need to Understand Monomers of Lipids?
Grasping the monomers of lipids is essential because these are the building blocks that form complex lipid structures. Knowing these fundamental components can help you understand how they are synthesized, how they interact with other cellular components, and how they fulfill critical functions in the body. Whether you are a student, a health professional, or simply a curious mind, this guide will help you navigate through the complexities of lipid monomers with clarity and ease.
Quick Reference
- Immediate action item: Identify common types of lipids such as triglycerides, phospholipids, and steroids.
- Essential tip: Learn how to distinguish between saturated and unsaturated fatty acids.
- Common mistake to avoid: Confusing lipid monomers with polymers; remember, monomers are simple building blocks.
Triglycerides: The Basic Lipid Monomers
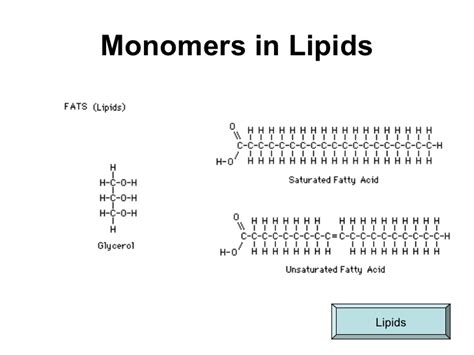
Triglycerides are one of the most common types of lipids found in the body and are the main constituents of body fat in humans and other animals. Understanding triglycerides starts with recognizing their basic structure.
A triglyceride is made up of three fatty acids attached to a glycerol backbone. These components are linked together through ester bonds, which are formed when a water molecule is released in a condensation reaction. The fatty acids can vary in length and saturation level, which influences the properties and functions of the triglyceride.
Here’s a detailed look at how triglycerides are formed:
- Glycerol Backbone: Triglycerides start with glycerol, a three-carbon alcohol that provides the backbone structure for the lipid molecule.
- Fatty Acids: Three fatty acids attach to the glycerol. These can be saturated (no double bonds between carbon atoms) or unsaturated (one or more double bonds between carbon atoms).
- Formation of Ester Bonds: The bond formation occurs when each hydroxyl group on the glycerol molecule reacts with the carboxyl group of a fatty acid, releasing a molecule of water for each ester bond formed.
Understanding this process helps you comprehend how the body stores and utilizes energy and how different types of fatty acids impact health.
Phospholipids: Key Players in Cellular Structure
Phospholipids are another major class of lipids that are critical for cellular function. Unlike triglycerides, phospholipids are amphipathic, meaning they have both hydrophilic (water-loving) and hydrophobic (water-repelling) parts. This characteristic makes them essential for forming cell membranes.
A phospholipid molecule consists of:
- Glycerol Backbone: Like triglycerides, phospholipids start with a glycerol molecule.
- Two Fatty Acids: Two fatty acids attach to the glycerol backbone, forming the hydrophobic “tail” regions.
- Phosphate Group: A phosphate group attaches to the third hydroxyl group of glycerol, forming the hydrophilic “head” of the phospholipid.
The phosphate group’s hydrophilic nature allows phospholipids to form bilayers in aqueous environments, creating a stable barrier that separates and protects cellular contents.
Steroids: Versatile Lipid Molecules
Steroids are another class of lipids that play diverse roles in the body. Unlike triglycerides and phospholipids, steroids are not formed from a glycerol backbone. Instead, they have a structure composed of four fused carbon rings.
Steroids are involved in various functions:
- Hormones: Many steroid hormones, such as estrogen and testosterone, regulate growth, reproduction, and metabolism.
- Membranes: Cholesterol, a type of sterol, is an integral component of cell membranes, affecting membrane fluidity and acting as a precursor to other steroids.
- Vitamin D: Another steroid, Vitamin D, plays a crucial role in calcium and phosphate metabolism, essential for bone health.
Quick Reference
- Immediate action item: Recognize that triglycerides are composed of glycerol and three fatty acids.
- Essential tip: Understand that phospholipids form the basic structure of cell membranes due to their amphipathic nature.
- Common mistake to avoid: Mistaking steroids for simple glycerol-based structures; remember their unique four-ring structure.
Practical Applications: How to Implement Your Knowledge
Now that you have a foundational understanding of lipid monomers, let’s explore how this knowledge can be applied in practical scenarios:
Application in Dietary Choices
Understanding lipid monomers helps in making informed dietary choices:
- Fat Intake: Knowing the difference between saturated and unsaturated fats can guide you in selecting healthier fats. For example, unsaturated fats found in nuts, seeds, and fish are generally better for heart health compared to saturated fats found in red meat and dairy products.
- Phospholipids: Foods rich in phospholipids, such as eggs and organ meats, can be beneficial for brain health.
- Steroids: While cholesterol is often stigmatized, it plays vital roles in the body, and its source (e.g., animal versus plant sources) can impact health differently.
Application in Scientific Research
For those involved in scientific research, understanding lipid monomers is fundamental:
- Cell Membrane Studies: Knowing the structure of phospholipids aids in understanding how membranes function and how they can be manipulated for research purposes.
- Hormonal Research: Detailed knowledge of steroid structures and functions can lead to breakthroughs in understanding and treating hormonal imbalances.
Practical FAQ: Answers to Common Questions
What is the difference between saturated and unsaturated fatty acids?
Saturated fatty acids have no double bonds between carbon atoms and are typically solid at room temperature. They are commonly found in animal fats like butter and lard. Unsaturated fatty acids have one or more double bonds, which create a bend in the molecule, making them generally liquid at room temperature. These are found in plant oils like olive and canola oil. The presence of double bonds in unsaturated fats makes them healthier options as they are associated with lower levels of bad cholesterol and heart disease risk.
Why are phospholipids important for cell membranes?
Phospholipids are crucial for cell membranes because of their amphipathic nature, which means they have both hydrophilic (water-attracting) and hydrophobic (water-repelling) parts. This allows them to form a bilayer that acts as a barrier, protecting the cell's internal environment while allowing certain substances to pass through. The hydrophilic heads face outward towards the aqueous environment, while the hydrophobic tails face inward, forming a stable and semi-permeable membrane.