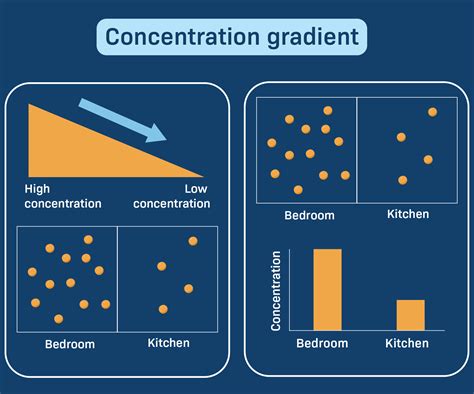
In the intricate dance of cellular processes, understanding what a concentration gradient means can greatly enhance your grasp of fundamental biology. A concentration gradient refers to the difference in the concentration of a substance between two points, often driving various biological functions. If you find yourself puzzled by this term or need actionable ways to understand it, you’ve come to the right place. This guide will provide you with the clarity you need, with a focus on practical, user-friendly solutions.
Understanding Concentration Gradients: A Problem-Solution Opening
The concept of a concentration gradient might sound daunting at first glance, but it’s a fundamental principle underlying many natural processes. From the diffusion of gases in the atmosphere to the movement of ions across cell membranes, understanding concentration gradients is crucial. However, many learners struggle with grasping this concept, leading to confusion and frustration. This guide aims to demystify concentration gradients, offering step-by-step guidance and actionable advice to make this scientific principle clear and accessible. Whether you're a student, a professional, or just curious, this guide will empower you to understand the nuances of concentration gradients, allowing you to apply this knowledge to solve real-world problems and advance your understanding of biological sciences.
Quick Reference
Quick Reference
- Immediate action item: Identify the two regions where the substance concentration differs. Focus on these regions to understand the gradient.
- Essential tip: Always remember that substances move from areas of higher concentration to lower concentration, seeking equilibrium.
- Common mistake to avoid: Do not confuse concentration gradient with speed of diffusion. The gradient drives the movement, but its rate can be affected by other factors like temperature and medium.
Detailed How-To Sections
What Is a Concentration Gradient?
A concentration gradient is essentially the difference in the concentration of a substance from one place to another. Imagine a room filled with a gas: if you release a balloon of perfume in one corner, the scent will disperse through the room. This dispersal happens because the concentration of the perfume is higher in the corner where the balloon was released and lower as it spreads out. This difference in concentration creates a gradient that pushes the perfume molecules to spread out evenly across the room.
How Concentration Gradients Drive Biological Processes
Biological processes are often driven by concentration gradients. For instance, the oxygen gradient in your lungs facilitates the diffusion of oxygen into your blood. Similarly, in cellular respiration, the glucose gradient inside the cell pushes the metabolic processes that convert glucose into energy. Understanding these gradients helps explain how cells function, how nutrients are transported, and how waste products are removed.
Practical Example: Diffusion Across a Semipermeable Membrane
Consider a semipermeable membrane separating two solutions with different concentrations of solute. The solute will move from an area of higher concentration to lower concentration across the membrane until equilibrium is reached. This movement is a direct result of the concentration gradient. For instance, in a dialysis machine used to treat kidney failure, a semipermeable membrane allows waste products to diffuse from the patient’s blood (high concentration) to a dialysate solution (lower concentration), cleansing the blood.
Step-by-Step Guide to Calculating Concentration Gradients
Calculating concentration gradients involves basic math. Here’s how you can do it:
- Step 1: Identify the concentrations of the substance at two different points. Let’s say you have two beakers: one with a concentration of 2 M (molar) and the other with 0.5 M.
- Step 2: Determine the difference between these two concentrations: 2 M - 0.5 M = 1.5 M.
- Step 3: Understand this difference as your concentration gradient, representing the driving force for diffusion or transport processes.
This simple calculation allows you to quantify the gradient, which is fundamental in understanding and predicting diffusion and transport phenomena.
Practical FAQ
How do concentration gradients affect cellular transport?
Concentration gradients are crucial for cellular transport, particularly in the movement of ions and molecules across cell membranes. Cells use semipermeable membranes that allow specific substances to pass through via processes such as osmosis, facilitated diffusion, and active transport. For example, water moves from an area of high water concentration (low solute concentration) to an area of low water concentration (high solute concentration) through osmosis. Similarly, glucose and other nutrients move across the membrane driven by their concentration gradients.
What happens when a concentration gradient is disrupted?
When a concentration gradient is disrupted, it can lead to various consequences depending on the context. For example, in a cell, if the gradient is disrupted, it can lead to loss of vital nutrients or accumulation of waste products, potentially impacting cellular functions. In industrial processes, disrupted gradients can affect the efficiency of separation techniques like chromatography or dialysis. In all cases, the disruption often means that natural processes driven by these gradients are either slowed down or halted until equilibrium is restored.
Best Practices for Understanding Concentration Gradients
To master the concept of concentration gradients, consider the following best practices:
- Visualize the Gradient: Use diagrams and visual aids to map out concentration differences across a space.
- Relate to Everyday Examples: Consider everyday scenarios, like the way heat distributes in a room or how coffee spreads in hot water, to grasp the principles of diffusion and gradient.
- Experiment and Observe: Conduct simple experiments, like mixing two solutions of different concentrations and observing the diffusion process over time.
- Study Real-World Applications: Look into real-world examples like atmospheric diffusion, osmosis in biological systems, or concentration gradients in chemical reactions to see these principles in action.
By following these best practices, you can deepen your understanding of concentration gradients and apply this knowledge effectively to both academic and real-world problems.
By systematically breaking down the concept of concentration gradients, and with practical examples, you now have a clearer understanding of how these principles operate in various contexts. From basic diffusion to complex cellular processes, grasping the concentration gradient can significantly enhance your comprehension of natural and biological sciences. Use the insights and practical tips provided here to navigate through more advanced topics and apply this knowledge effectively to your academic or professional endeavors.