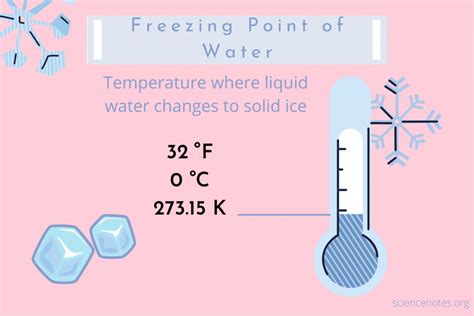
Understanding the exact freezing temperature in Fahrenheit is crucial for numerous applications ranging from scientific research to everyday life. Water freezing marks a transition from liquid to solid state at a specific point, which in Fahrenheit is precisely 32°F. This seemingly straightforward fact becomes pivotal when precise measurements are required, whether you are conducting experiments in chemistry or simply planning outdoor activities for the winter season.
This article delves into the significance of knowing the exact freezing temperature in Fahrenheit and provides practical insights to help you grasp this fundamental concept. We’ll explore how this knowledge can be applied in real-world scenarios, highlight the importance of accuracy, and present expert tips to measure temperature effectively.
Key Insights
- Water freezes at 32°F, a fundamental fact that finds numerous applications.
- Precision in measurement is crucial for scientific research and practical applications.
- Utilize accurate thermometers and understand calibration techniques for precise readings.
Water’s transition to ice at 32°F is not just a theoretical concept but a critical parameter for many practical situations. In the realm of chemistry, accurate temperature readings ensure the integrity of experiments involving endothermic and exothermic reactions. For instance, a slight deviation in the freezing point can alter reaction rates and outcomes, which can be detrimental in drug formulation processes.
In the field of meteorology, the exact freezing temperature is vital for accurate weather forecasting. Forecasts often depend on precise temperature measurements to predict frost, ice formation, and other winter weather phenomena. These predictions are crucial for public safety, helping to plan for road conditions, flight schedules, and even agricultural practices.
The scientific and practical importance of knowing the exact freezing temperature in Fahrenheit highlights the need for precision. Understanding this threshold allows for better management of resources and safer outcomes in various activities, including preserving perishable goods and ensuring infrastructure resilience during winter.
Why is it important to know the exact freezing temperature?
Knowing the exact freezing temperature of water is important for scientific accuracy, weather forecasting, safety in outdoor activities, and preserving perishable goods. Precision in measurements ensures better outcomes in research, public safety, and day-to-day operations.
How can I measure the freezing temperature accurately?
To measure the freezing temperature accurately, use calibrated digital or mercury thermometers. Ensure the thermometer is free of air bubbles, immerse it in a water sample, and wait until the temperature stabilizes. For the highest accuracy, consider using a professional-grade lab thermometer or a digital one with calibration features.
This guide underscores the significance of knowing the exact freezing temperature in Fahrenheit. By leveraging this knowledge, you can enhance scientific research accuracy, improve weather prediction models, and make safer decisions in daily activities. It’s important to utilize precise instruments and understand the principles of calibration to ensure the accuracy of your temperature readings. This expertise not only benefits your professional endeavors but also enriches everyday decision-making processes.