Understanding the freezing point of water is not just an academic exercise; it's a concept with practical relevance in everyday life, from managing food storage to understanding weather patterns. This guide dives into the fundamental principles behind the freezing point of water and offers you actionable advice for applying this knowledge effectively. We'll explore real-world examples and practical solutions to common problems related to water's freezing point. Whether you're a curious mind looking to grasp scientific concepts or someone facing challenges related to water temperature, this guide will serve as your reliable companion.
Introduction to Water’s Freezing Point
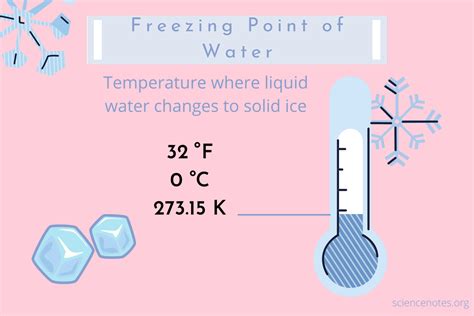
Water freezes at 32 degrees Fahrenheit (0 degrees Celsius) under standard atmospheric pressure. This pivotal temperature is where liquid water transforms into ice, a phase change crucial for both natural and industrial processes. Beyond basic scientific interest, the freezing point of water plays a vital role in preserving perishable goods, understanding climate change, and even in the survival of aquatic life during winter months.
Why Knowing Water’s Freezing Point Matters
Understanding water’s freezing point can help in various practical scenarios:
- Food preservation: Knowing when water freezes helps in properly storing food to prevent spoilage.
- Climate observation: It’s integral to predicting weather patterns and understanding climate change impacts.
- Survival skills: For outdoor enthusiasts or in survival situations, knowing the freezing point aids in making informed decisions about water and food storage.
Quick Reference
Quick Reference
- Immediate action item: Keep your freezer at 0°F (-18°C) to ensure food is preserved effectively.
- Essential tip: Use ice packs that are at or below 32°F (0°C) for quick cooling of perishable items.
- Common mistake to avoid: Overlooking the effect of altitude on water’s freezing point can lead to incorrect assumptions about when and how water will freeze.
The Science Behind Water’s Freezing Point
To appreciate the freezing point of water, it’s important to delve into the science that explains why water turns to ice at 32°F (0°C) under standard atmospheric pressure. Water molecules are polar, meaning they have a slight positive charge on one end and a slight negative charge on the other. These charges create an attraction between water molecules, leading to the formation of a hydrogen bond network in liquid water.
When water cools down to its freezing point, the kinetic energy of the water molecules decreases. At 32°F, the thermal energy is low enough for the hydrogen bonds to form a crystalline structure, creating ice. This phase change is exothermic, meaning it releases energy as the water transitions from liquid to solid.
Practical Application: Ensuring Food Preservation
Food preservation is one of the most direct applications of understanding the freezing point of water. Proper understanding and application can prevent food waste and ensure nutritional safety.
Here’s a detailed approach to ensure your food is preserved correctly:
Step-by-Step Guide to Effective Food Freezing
Freezing food not only extends its shelf life but also allows you to enjoy meals without the need for constant refrigeration. Here’s how to do it right:
- Preparation: Before freezing, ensure that your food is properly cooled. If food is warm when placed in the freezer, it may thaw and refreeze, leading to texture and quality deterioration.
- Packaging: Use airtight, freezer-safe containers or bags to prevent freezer burn. This not only preserves the quality of the food but also saves space in your freezer.
- Labeling: Always label your frozen food with the contents and the date. This helps you keep track of what you have and prioritizes freezing new items to replace older ones.
- Storage: Place new items in the front of the freezer to ensure they are the first to thaw when you need to use them. This practice helps maintain the optimal freezing temperature of the older items.
Practical Example: Freezing Leftover Meals
Imagine you’ve cooked a large meal and have leftovers. Here’s how to freeze them effectively:
- Cool the leftovers completely to room temperature before freezing.
- Divide the meal into meal-sized portions using freezer-safe containers or bags.
- Seal the containers or bags tightly and remove as much air as possible from the bags before sealing.
- Label each container or bag with the contents and the date, and store it in the front of the freezer.
FAQ Section
What’s the best temperature to keep my freezer for optimal food preservation?
For optimal food preservation, it’s best to keep your freezer at a consistent temperature of 0°F (-18°C). This ensures that the food remains frozen solid, preventing any thawing or refreezing cycles that could affect the quality and safety of the food.
Advanced Tips: Exploring Altitude’s Effect on Freezing Point
While 32°F (0°C) is the freezing point under standard atmospheric pressure, altitude can alter this slightly. As you increase in altitude, atmospheric pressure decreases, which can lower the freezing point slightly. This is why bodies of water in high altitude regions may freeze at slightly lower temperatures. For most practical purposes, unless you’re at extreme altitudes or dealing with pure scientific research, this deviation is minimal and can usually be overlooked.
However, understanding this principle is crucial for specific applications like mountain region water storage or understanding climates.
Conclusion
Understanding the freezing point of water is more than just a scientific curiosity; it’s a practical skill with wide-ranging applications from food preservation to climate observation. By delving into the science and applying practical advice, you can enhance your daily life and ensure that you’re making informed decisions based on a solid understanding of this fundamental property of water.
Remember, the key to mastery lies in practice and application. So, whether you’re storing food, planning outdoor adventures, or simply curious about the natural world, applying the knowledge of water’s freezing point can offer you a wealth of practical benefits.