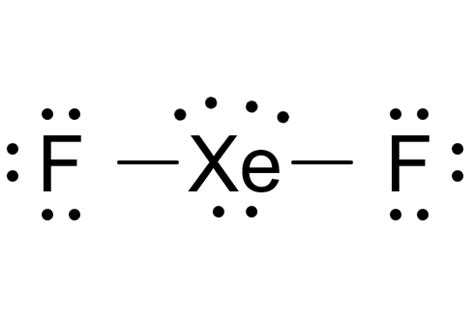Understanding the XeF2 Lewis Structure: A Clear and Authoritative Guide
When delving into the world of molecular chemistry, it’s imperative to have a firm grasp on Lewis structures to fully comprehend molecular geometry and bonding. The XeF2 Lewis structure is an excellent case study due to its unique arrangement and the xenon atom’s expanded octet configuration. This exploration will provide practical insights, backed by evidence, to demystify the complexities of this structure.
Essential Background: What is the XeF2 Lewis Structure?
The XeF2 molecule comprises a central xenon atom bonded to two fluorine atoms. Xenon, a noble gas, traditionally has a full octet, but in this molecule, it utilizes its available d-orbitals to accommodate more than eight electrons—an example of an expanded octet. This unique configuration allows us to understand how xenon can form stable molecules beyond its usual configuration.
Key Insights
- The primary insight is understanding xenon’s ability to expand its octet, allowing it to form two bonds while maintaining stability.
- A technical consideration is the placement of lone pairs which impacts the molecule’s geometry and overall stability.
- An actionable recommendation is to use the VSEPR theory to predict the molecular geometry around the central atom.
Visual Representation and Electron Placement
In drawing the XeF2 Lewis structure, it’s crucial to distribute electrons correctly. Begin by placing the xenon atom at the center, surrounded by two fluorine atoms. Xenon will use its five valence electrons to bond with two fluorine atoms, each of which requires one electron to complete their outer shell. Each fluorine atom shares one electron with xenon, resulting in two single bonds.
Following the octet rule for fluorine is crucial; however, for xenon, an expanded octet must be considered. Xenon, despite forming two bonds, has three lone pairs surrounding it. Each lone pair occupies two electron positions. The presence of these lone pairs is pivotal as they influence the molecule’s geometry, leading to a linear shape with a bond angle of 180 degrees, according to VSEPR theory.
Understanding Molecular Geometry
The XeF2 molecule’s geometry is a linear shape, which might seem counterintuitive given the three lone pairs around xenon. To comprehend this, one must revisit the principles of the VSEPR theory. The theory predicts molecular geometry based on minimizing electron repulsions. With three lone pairs and two bonded pairs of electrons, the lone pairs will occupy positions as far apart as possible, thus forming a linear structure.
When we apply the VSEPR theory, the lone pairs, by occupying axial positions, push the fluorine atoms outward, ensuring the molecule retains its linear structure. This understanding is crucial for visualizing and predicting the behavior of similar molecules.
Why does xenon in XeF2 have an expanded octet?
Xenon in the XeF2 molecule exhibits an expanded octet because it has available d-orbitals. Unlike second-period elements, xenon can use these d-orbitals to hold more than eight electrons, allowing it to form two bonds with fluorine atoms while maintaining stability.
How does the geometry of XeF2 impact its reactivity?
The linear geometry of XeF2 minimizes repulsion between electron pairs around the xenon atom, resulting in a stable molecule. This stability contributes to the molecule’s relatively low reactivity under standard conditions. However, it can still participate in reactions that disrupt its structure, such as when it reacts with strong bases.
This comprehensive analysis sheds light on the intricate details of the XeF2 Lewis structure, providing a foundation for further exploration into the fascinating field of molecular chemistry. By understanding these principles, we gain deeper insights into the behavior and reactivity of molecules beyond the basics.