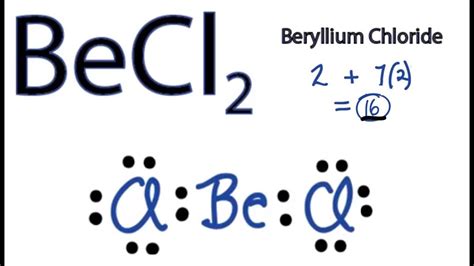The exploration of BeCl2 Lewis structure offers a critical gateway into understanding the foundational principles of chemistry and molecular bonding. The BeCl2 molecule epitomizes the beauty of covalent bonding and the simplicity of valence shell electron pair repulsion (VSEPR) theory. To unravel this fascinating structure, we delve deep into the atomic behavior, molecular geometry, and the electronic configuration of beryllium and chlorine atoms.
Key Insights
- Beryllium chloride (BeCl2) showcases the principles of molecular geometry and Lewis structures.
- Understanding the electronic configuration of beryllium and chlorine aids in determining the molecule's shape.
- A linear geometry is expected for BeCl2 due to the electron pair repulsion theory.
Beryllium Chloride’s Electron Configuration
In the realm of quantum chemistry, the electronic configuration holds the key to molecular behavior. Beryllium, with an atomic number of 4, has the electron configuration of 1s² 2s². When forming BeCl2, beryllium loses both of its outer electrons to achieve a noble gas configuration, yielding a +2 oxidation state. Each chlorine atom, with an atomic number of 17 and the configuration of [Ne] 3s² 3p⁵, gains one electron to fulfill its octet rule, resulting in a -1 charge. Consequently, beryllium forms two covalent bonds with two chlorine atoms, balancing the charge through these connections.VSEPR Theory Application to BeCl2
Valence Shell Electron Pair Repulsion (VSEPR) theory is paramount in predicting the spatial arrangement of atoms in molecules. For BeCl2, the central beryllium atom shares electrons with two chlorine atoms, resulting in two regions of electron density around it. VSEPR theory posits that these regions will arrange themselves to minimize repulsion, leading to a linear geometry. Thus, in BeCl2, the two chloride ions positioned at 180 degrees to each other optimize the electron pair repulsion, maintaining a stable linear structure.FAQ Section
What is the hybridization of the beryllium atom in BeCl2?
In BeCl2, the beryllium atom undergoes sp hybridization. This hybridization allows the formation of two sp hybrid orbitals, each of which overlaps with the p orbital of each chlorine atom, resulting in the linear geometry of the molecule.
Why does BeCl2 have a linear shape?
BeCl2 is linear because it has only two bonding pairs of electrons around the beryllium atom, and VSEPR theory dictates that two electron pairs will arrange themselves to be as far apart as possible. This results in a 180-degree bond angle and a linear molecular shape.
This deep dive into BeCl2’s Lewis structure and its molecular geometry underscores the elegance of chemical bonding and molecular architecture. With its linear structure dictated by the VSEPR theory and the electron pair repulsion principle, BeCl2 serves as an exemplar for comprehending the foundational aspects of molecular chemistry.