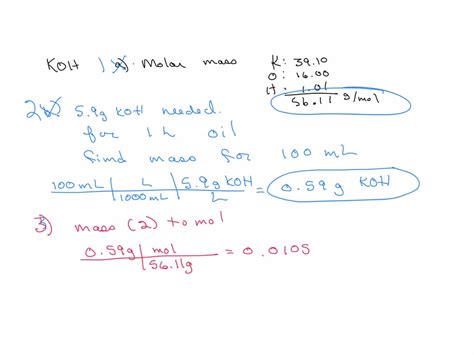Understanding the molar mass of ethanol is essential for chemists and hobbyists alike, whether for basic science experiments, brewing, or chemical analysis. Knowing this fundamental property can help you to accurately measure, mix, and convert between different units in various chemical calculations. This guide will provide step-by-step guidance with actionable advice, real-world examples, and practical solutions to empower you to master ethanol’s molar mass with confidence and ease.
Understanding the Importance of Ethanol Molar Mass
Ethanol, also known as ethyl alcohol (C2H5OH), is a widely used compound in many industries, including beverages, fuel, and chemical synthesis. Knowing the molar mass of ethanol—the weight of one mole of ethanol molecules—is crucial for a multitude of applications, including chemical reactions, fermentation processes, and analytical chemistry. Mastering this concept will provide a solid foundation for more advanced scientific pursuits.
Problem-Solution Opening: The Challenge and Solution
One of the common challenges many users face is understanding how to calculate the molar mass of ethanol accurately. This can lead to mistakes in chemical calculations, which can have serious consequences in laboratory settings. The solution lies in a step-by-step approach to calculating the molar mass of ethanol. This guide will break down the process into simple, actionable steps, replete with practical examples and real-world applications to help you grasp the concept thoroughly.
Quick Reference
Quick Reference
- Immediate action item with clear benefit: Calculate ethanol’s molar mass using its chemical formula, ensuring accuracy for all your chemical calculations.
- Essential tip with step-by-step guidance: Break down ethanol’s molecular structure to understand how to sum the atomic masses of each element.
- Common mistake to avoid with solution: Confusing ethanol’s molar mass with other compounds’ molar masses; always ensure you are using ethanol’s specific formula (C2H5OH) when calculating.
How to Calculate Ethanol’s Molar Mass
The molar mass is a measure of the total mass of one mole of a substance. To find the molar mass of ethanol (C2H5OH), you need to sum the atomic masses of all the atoms in one molecule of ethanol.
Here’s a detailed breakdown:
- Start with ethanol's chemical formula: C2H5OH. This means one molecule of ethanol contains:
- 2 carbon (C) atoms
- 6 hydrogen (H) atoms
- 1 oxygen (O) atom
- Find the atomic masses of each element from the periodic table:
- Carbon (C): approximately 12.01 grams per mole
- Hydrogen (H): approximately 1.01 grams per mole
- Oxygen (O): approximately 16.00 grams per mole
- Calculate the molar mass of ethanol by summing the total atomic masses:
- For Carbon: 2 × 12.01 g/mol = 24.02 g/mol
- For Hydrogen: 6 × 1.01 g/mol = 6.06 g/mol
- For Oxygen: 1 × 16.00 g/mol = 16.00 g/mol
- Add these together:
- Total molar mass of ethanol = 24.02 g/mol (C) + 6.06 g/mol (H) + 16.00 g/mol (O)
- Total molar mass of ethanol = 46.08 g/mol
This comprehensive step-by-step process will ensure that you accurately determine ethanol’s molar mass, avoiding errors in chemical calculations.
Real-World Application of Ethanol’s Molar Mass
Understanding ethanol’s molar mass is crucial for various practical applications. For instance, in the brewing industry, precise measurements and calculations are essential for the fermentation process. Similarly, chemists need accurate molar mass information for creating solutions, performing titrations, and synthesizing new compounds.
To illustrate, consider a scenario where a brewer is creating a new beer recipe. They need to calculate the amount of ethanol needed to achieve a desired alcohol by volume (ABV). Knowing ethanol's molar mass allows them to convert volume measurements to mass measurements, ensuring consistency and quality in the final product.
Practical Examples and Exercises
Here are a few practical examples to further solidify your understanding of ethanol’s molar mass:
- Example 1: Suppose you want to determine how much ethanol (in grams) is present in 1 liter of an 80% ethanol solution. First, calculate the molar mass of ethanol (46.08 g/mol), then use the percentage concentration to determine the mass. 80% ethanol means 80 grams of ethanol per 100 mL of solution. So, for 1 liter (1000 mL):
- 80 grams/100 mL × 1000 mL = 800 grams of ethanol in 1 liter of solution
- Example 2: If you have 0.5 moles of ethanol, calculate the mass:
- Mass = molar mass × number of moles = 46.08 g/mol × 0.5 moles = 23.04 grams
Common FAQ
Why is ethanol’s molar mass important in scientific calculations?
Ethanol’s molar mass is crucial for accurately determining the quantity of ethanol in a given solution. This is essential for various applications such as fermentation, chemical synthesis, and analytical procedures. It ensures precise measurements and successful outcomes in laboratory and industrial processes.
How does the molar mass of ethanol affect the density of its solutions?
The molar mass of ethanol directly impacts the density of ethanol solutions. Since density is defined as mass per unit volume, knowing ethanol’s molar mass allows you to calculate the density of ethanol solutions accurately. This is vital for applications where precise densities are needed, such as creating specific concentrations in chemical reactions.
Advanced Applications and Tips
Once you’ve mastered the basic calculation, here are some advanced applications and tips to take your understanding to the next level:
- Reactivity studies: Knowing the molar mass can help in determining the stoichiometry of ethanol in reactions, ensuring accurate reagent quantities.
- Industrial synthesis: In industrial settings, precise molar mass calculations are essential for synthesizing ethanol-based products, ensuring consistent product quality.
- Environmental analysis: In environmental studies, understanding ethanol’s molar mass is crucial for quantifying ethanol in air or water samples, contributing to environmental monitoring and safety assessments.
To keep your understanding sharp, consider these best practices:
- Practice: Regularly calculate the molar mass of different compounds to reinforce your skills.
- Use tools: Employ calculators and software for complex calculations to verify your results.
- Stay updated: Keep abreast of updates in atomic mass values from reliable sources, as periodic updates may occur.
By following this guide, you will gain a comprehensive understanding of ethanol’s molar mass, enabling you to tackle a variety of practical and advanced applications with confidence and precision. Happy calculating!