To grasp the concept of the molar mass of octane and understand its significance in various scientific and industrial applications, you need a practical, step-by-step guide. This guide is here to walk you through not only how to calculate the molar mass but also to address the challenges and misconceptions users often encounter. Whether you are a chemistry student, a chemical engineer, or simply curious about the precise nature of molecular weights, this guide will serve as a valuable resource.
Understanding the Importance of Octane’s Molar Mass
The molar mass of a compound like octane is crucial in fields such as chemistry, chemical engineering, and related sciences. It allows chemists and engineers to understand and predict the behavior of octane in various reactions and processes. For instance, knowing the molar mass of octane is essential for stoichiometric calculations, where the precise measurement of reactants and products in chemical reactions is critical. Additionally, it is used in determining the density, volume, and mass relationships, and plays a role in the study of hydrocarbons, which are fundamental in the production of gasoline and other fuels.
Quick Reference
Quick Reference
- Immediate action item: Calculate the molar mass of octane using its molecular formula, C8H18, and know that carbon © has an atomic mass of approximately 12.01 g/mol, and hydrogen (H) has an atomic mass of approximately 1.008 g/mol.
- Essential tip: Break down the calculation into manageable steps. Count the number of carbon and hydrogen atoms, multiply by their respective atomic masses, and add them together to get the molar mass of octane.
- Common mistake to avoid: Confusing atomic mass with molecular mass or forgetting to account for all hydrogen atoms in the formula.
How to Calculate the Molar Mass of Octane
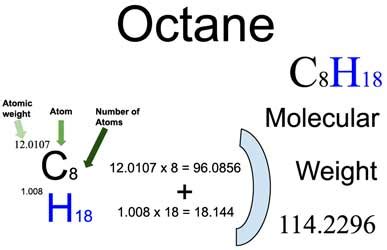
To calculate the molar mass of octane, you need to start with its molecular formula: C8H18. Follow these steps to understand and apply the calculation:
Step 1: Identify the Molecular Formula
Octane’s molecular formula is C8H18. This means the molecule consists of 8 carbon © atoms and 18 hydrogen (H) atoms. Understanding the molecular formula is the first crucial step in determining the molar mass.
Step 2: Determine Atomic Masses
You need to know the atomic masses of carbon and hydrogen, which are approximately:
- Carbon ©: 12.01 g/mol
- Hydrogen (H): 1.008 g/mol
These atomic masses will be used in the next steps to calculate the molar mass.
Step 3: Multiply Atomic Masses by Number of Atoms
For each type of atom in octane, multiply the atomic mass by the number of that type of atom in the molecule:
- For carbon: 12.01 g/mol x 8 = 96.08 g/mol
- For hydrogen: 1.008 g/mol x 18 = 18.144 g/mol
Step 4: Add the Masses Together
Add the masses obtained in step 3 to get the total molar mass of octane:
96.08 g/mol (carbon) + 18.144 g/mol (hydrogen) = 114.224 g/mol
Therefore, the molar mass of octane is 114.224 g/mol.
Step 5: Verify Your Calculation
To ensure accuracy, verify your calculation by checking each step for arithmetic errors and confirming the use of correct atomic masses from a reliable source such as the periodic table.
Practical Example: Using Molar Mass in Stoichiometric Calculations
Let’s explore a practical application of octane’s molar mass in a stoichiometric calculation. Suppose you need to determine how many moles of octane are present in a 1 kg sample:
Step 1: Convert Mass to Grams
Since the sample is 1 kg, convert it to grams because molar mass is in g/mol:
1 kg = 1000 grams
Step 2: Use the Formula to Find Moles
Use the formula:
Number of moles = mass (g) / molar mass (g/mol)
Number of moles of octane = 1000 g / 114.224 g/mol = 8.74 moles
Thus, there are 8.74 moles of octane in the 1 kg sample.
Practical FAQ
Why is knowing the molar mass of octane important?
Knowing the molar mass of octane is crucial for several reasons:
- It helps in understanding the weight and volume relationships in chemical reactions.
- It aids in stoichiometric calculations, which are essential for determining the amount of reactants needed and products formed in chemical reactions.
- It is used in fields like fuel production and chemical engineering to design and optimize processes.
- It also plays a role in environmental studies and assessments where the behavior and impact of hydrocarbons are analyzed.
Tips and Best Practices
Here are some additional tips and best practices to keep in mind when working with molar mass calculations:
- Use reliable atomic mass values: Always ensure that you are using the most up-to-date and accurate atomic mass values from a reliable source.
- Double-check your work: It’s easy to make a mistake in calculations. Always double-check your work by going through each step carefully.
- Practice with different compounds: To become more proficient in molar mass calculations, practice with different molecular formulas to gain confidence and accuracy.
- Understand the units: Make sure to use consistent units (grams and grams per mole) throughout your calculations to avoid errors.
Advanced Application: Octane in Fuel Blending
For chemical engineers and scientists working on fuel blending, understanding the molar mass of octane is critical. It helps in precisely calculating the proportion of octane needed to achieve a specific energy output and performance characteristic in fuels. Here’s how you can apply it:
Step 1: Determine Desired Octane Content
Identify the desired octane rating for your fuel blend, often measured in terms of Research Octane Number (RON) or Motor Octane Number (MON).
Step 2: Calculate Required Volume or Mass
To determine the required volume or mass of octane to add to your fuel blend, you will need to know the total volume or mass of the fuel mixture and the specific percentage of octane required:
Required mass of octane (g) = (Desired percentage of octane / 100) x Total mass of fuel blend (g)
Required volume of octane (L) = (Desired percentage of octane / 100) x Total volume of fuel blend (L)
Step 4: Convert to Usable Units
Use the molar mass of octane to convert between mass and volume if necessary, ensuring precise measurements:
For example, if you need to find out how many liters of octane at standard temperature and pressure (STP) are needed to achieve a certain mass, use the ideal gas law or appropriate conversion factors.
In conclusion, mastering the calculation of the molar mass of octane is not only academically rewarding but also practically indispensable for various scientific and industrial applications. By following the steps outlined in